-
近年来,淡水危机日益严重,非常规水资源制备淡水是解决淡水危机的重要途径之一[1]。目前应用较为广泛的淡水制备工艺有电渗析(electrodialysis,ED)、反渗透(reverse osmosis,RO)和纳滤(nanofiltration,NF),但这些工艺普遍存在能耗高、操作复杂且具有二次污染的风险[2]。近年来,流动电极电容去离子(flow electrode capacitive deionization,FCDI)作为一种新型的脱盐技术,具有能耗低、操作简单、绿色无污染等优点而备受关注[3]。
FCDI技术是由电容去离子技术(capacitive deionization,CDI)衍变而来,在CDI装置的基础上引入离子交换膜和流动电极,其原理是通过在电极上施加电压,将进水中的盐离子吸附在电极微孔与溶液界面的双电层(electric double layer,EDL)上,从而达到脱盐的目的[4-5]。通过在两侧电极与中间腔室间加入离子交换膜能够有效的抑制CDI装置中的同离子效应,显著提高了其电荷效率,电荷效率可由60%左右提升至85%[6]。此外,流动电极相比固体电极操作模式更加灵活,能够通过调节电极循环方式实现电极在吸附的同时完成再生,克服了固体电极吸附容量低,无法连续运行的问题[7]。
FCDI技术中所用到的流动电极由电极材料、导电剂和流体介质组成,如何提高流动电极的导电性和电极材料对离子的吸附性能一直受到研究者们的广泛研究。其中,对电极材料进行掺杂改性是提高流动电极的电化学性能的有效手段。ALSULTAN等[8]和YAN等[9]分别以经过直接蓝(direct blue,DB)分子修饰和硝酸、对氨基苯磺酸磺化改性后的炭黑(carbon black CB)和碳纳米管(carbon nanotubes CNT)为电极材料,均有效解决了其由疏水性导致的团聚和沉降问题,显著提升了系统的平均脱盐速率,其中磺化CNT流动电极对比CNT流动电极平均脱盐速率提升了74.6%。但上述研究中对改性材料的需求量较高,往往需要较高的成本,且会对环境造成二次污染[10]。有研究表明,在流动电极中添加具有强导电性的碳材料作为导电剂,能够在电极材料之间建立电荷传输通道,提高流动电极的导电性和离子吸附性能。LIANG等[11]和CHO等[12]以活性炭(activated carbon, AC)作为电极材料并分别将CB和CNT作为导电剂投加到流动电极中,均有效地提高了流动电极的脱盐性能。但CB、CNTs的高疏水性会导致其难以在流动电极中均匀分布,不仅影响导电效果,还会显著增加流动电极的粘度,增加泵送能耗[13]。有研究表明,在碳材料表面引入含氧官能团能够改善其疏水性,增加比电容,进而提升了其对离子的吸附性能[14-15]。H2O2作为一种常用的氧化剂,有易分解、无过量氧化剂残留与二次污染风险等优点,这些优势为H2O2对导电剂氧化改性的应用提供了可能。因此,探究工艺简单、绿色无污染的导电剂氧化技术对于推进FCDI在脱盐领域的发展势在必行。
本论文利用H2O2氧化CB,对氧化后的CB进行了物理和电化学特性表征,并考察了其作为导电剂添加到流动电极中,FCDI装置的脱盐性能,进一步探讨了在FCDI体系中提升流动电极脱盐性能方面的机制,并考察了CB氧化程度、运行电压、投加量对FCDI系统脱盐性能的影响,以期为后期工程化应用提供参考。
-
取1 g CB(阿法埃莎(中国)化学有限公司 H30253)浸泡在100 mL(30%)的双氧水(天津市永大化学试剂有限公司)中,室温下分别搅拌0、6、12、24 h。然后通过抽滤将CB分离,并用去离子水充分洗涤,80 ℃烘箱中干燥6 h。最后,用研钵将烘干后的CB充分研磨,按照氧化时间分别命名为CBO-0、CBO-6、CBO-12、CBO-24。
-
将AC(福州益环碳素有限公司 YEC-200D)、导电剂(CB或CBO)分别以质量比为1.5%和0.25%的比例与0.6 g∙L−1的NaCl溶液在磁力搅拌器上搅拌2 h。得到的质地均匀的碳浆即为本实验中所使用的流动电极。此外,导电剂含量为0.1%、0.5%的流动电极配制方法同上,其中仅调整导电剂的添加量。
-
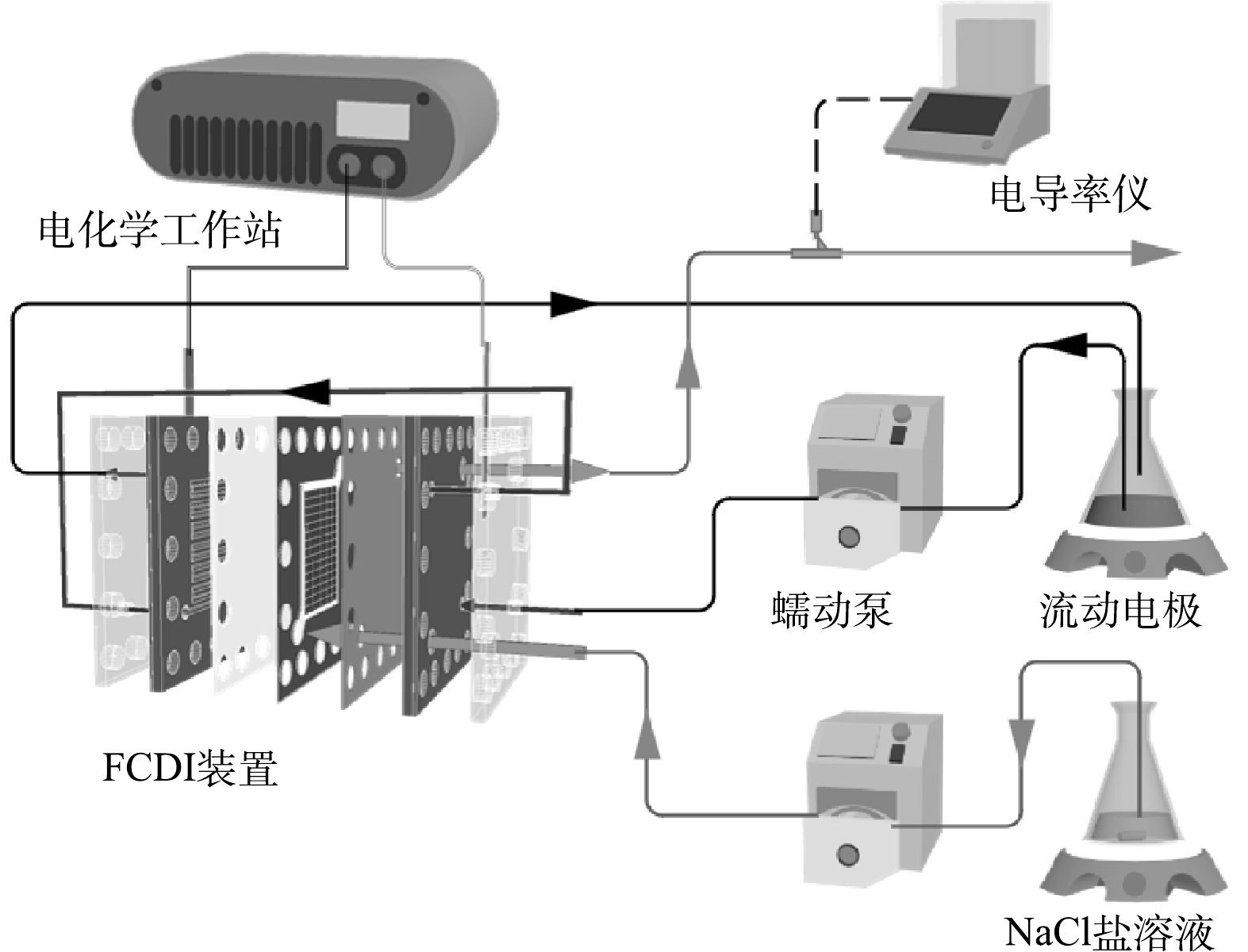
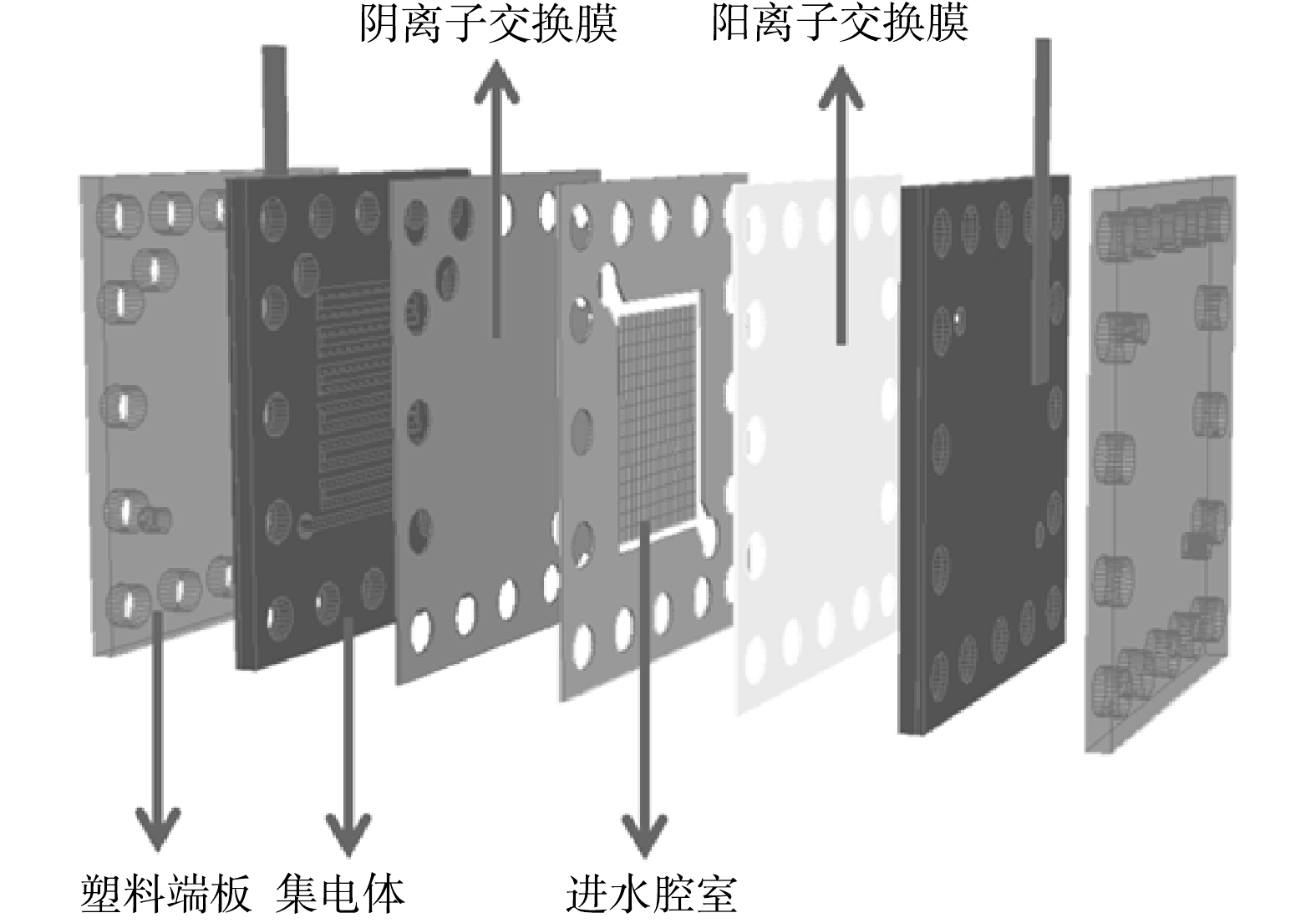
如图1所示,FCDI装置由塑料端板、集电体、阳离子交换膜(CEM-DF-120,山东天维膜科技有限公司)、阴离子交换膜(AEM-DF-120,山东天维膜科技有限公司)和由硅胶垫和塑料格网组成的中间腔室组成,并在两组件之间加入硅胶垫片密封。在单次脱盐实验中,取40 mL流动电极并以单循环(single-cycle,SC)模式在集电体中以10 ml∙min−1的流速运行[16],由阴极侧的集电体中流出后直接流入阳极侧的集电体,最后流出装置返回电极储存瓶中。
-
本文所用的脱盐系统如图2所示,该系统由蠕动泵(BT100-1L,保定兰格恒流源有限公司)、电导率仪(S475-UMIX,梅特勒Seven Excellence)、FCDI脱盐装置和电化学工作站(瑞士万通,MAC90589)组成。进水为模拟盐溶液0.6 g∙L−1 NaCl,并以连续流模式2.4 mL min−1的流速流经系统。本文采用单一变量法分别对应用不同氧化程度CBO(CBO-0、CBO-6、CBO-12、CBO-24)、不同电压(0.8、1.2、1.6 V)和不同投加量(0.1%、0.25%、0.5%)时系统的脱盐性能进行探究。单次脱盐实验加电900 s,期间用电导率仪实时监测出水电导率。
系统的脱盐性能通过脱盐率(salt removal rate,SRR)、电荷效率(charge efficiency,CE)、平均脱盐速度(average salt removal rate,ASRR)和单位能量脱盐量(energy normalization removal salt,ENRS)进行评估。脱盐率、平均脱盐速度、电荷效率、单位能量脱盐量分别根据式(1)、式(2)、式(3)、式(4)计算。
式中:η为脱盐率,%;C0为进水盐溶液的摩尔浓度,g∙L−1;Ct为脱盐t时刻溶液的物质的量浓度,g∙L−1;t为吸附时间,s。
式中:α为平均脱盐速度,μmol∙(cm2∙min)−1;S为流动电极与离子交换膜的接触面积,cm2。
式中:ρ为电荷效率,%;∫Idt为吸附阶段消耗的电荷量,C。
式中:β为单位能量脱盐量,μmol∙J−1;V为FCDI装置的施加电压,V。
-
本研究通过分别对CBO-0、CBO-6、CBO-12和CBO-24进行扫描电子显微镜(SEM,捷克TESCAN MIRA LMS)、氮气吸附/脱附技术(BET,美国Micromeritics ASAP 2460)、X射线光电子能谱(XPS,美国Thermo Scientific K-Alpha)和接触角/表面张力测量(承德鼎盛JY-82C视频接触角测定仪)四种物理表征来观察随着氧化时间的增长,CB的表面结构、孔隙结构、表面官能团含量和亲水性的变化趋势。其中,接触角测量采用的测量方法为座滴法,接触介质为去离子水。
-
本研究通过对以CBO-0、CBO-6、CBO-12和CBO-24为导电剂的流动电极进行循环伏安法(CV,瑞士万通 MAC90589)和电化学阻抗测试(EIS,瑞士万通 MAC90589)表征以观察流动电极电容和阻抗的变化。测试环境为双电极体系的密闭电解池。CV扫描电压为±1.2 V,扫速50 mV∙s−1。EIS扫描频率为0.1~105 Hz。CV曲线的比电容根据式(5)进行计算。
式中:C为比电容,F∙g−1;a为扫速,mV∙s−1;m为碳材料质量,g;Va和Vc分别为扫描电压的低电位截止值和高电位截止值,V。
-
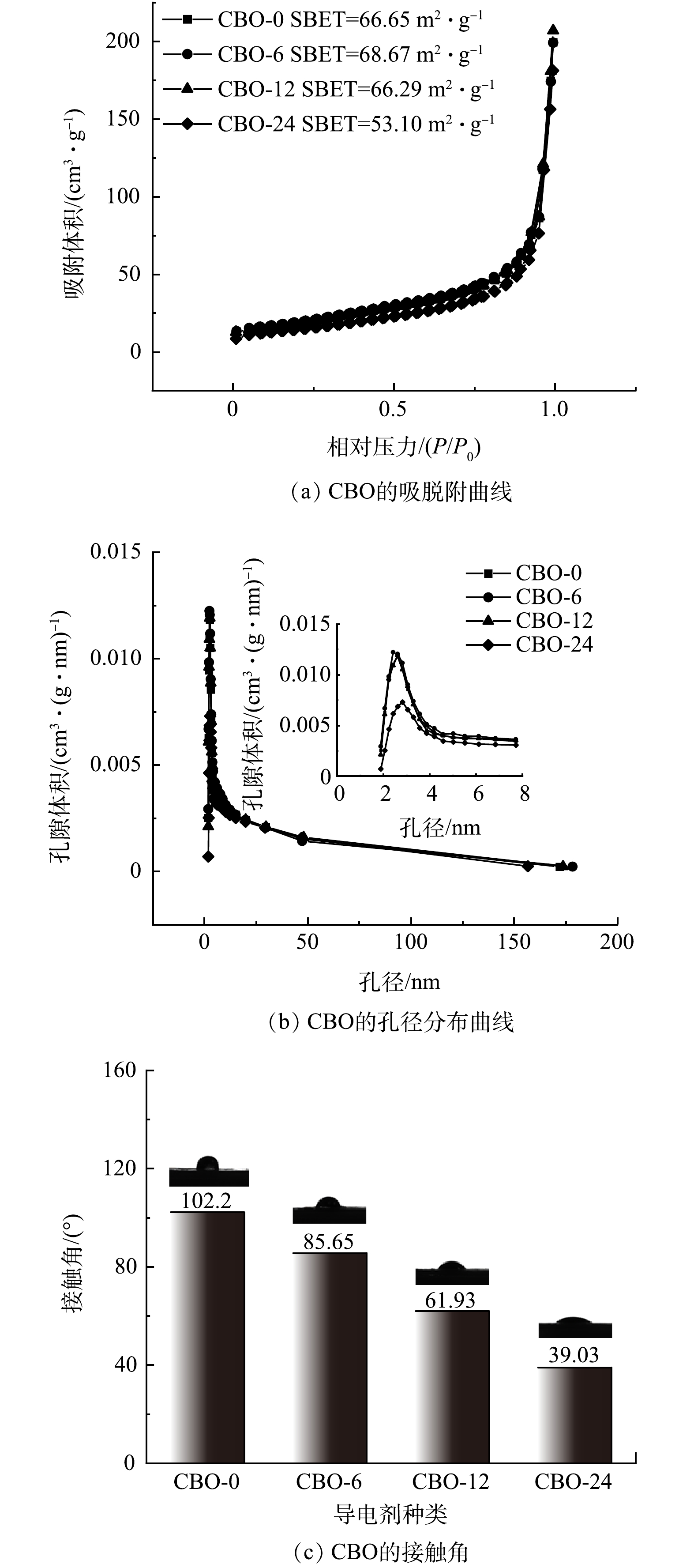
由图3(a)可以看出,CBO-0由形状规则的颗粒团聚组成,团聚体的直径约为300 nm。经H2O2氧化后,CBO表面形态如图3(b)~(d)所示。可以看到,随着氧化时间的增长,CB表面的炭黑粒子的团聚程度愈发紧密,团聚体的直径也明显升高。这可能是由于氧化改性为CB表面提供的C—O、C=O等含氧官能团,从而改变了炭黑粒子的表面活性,增强了炭黑粒子间的相互吸引力。
由图4(a)可以看出,CBO-0、CBO-6和CBO-12的比表面积非常接近,这表明适当的氧化处理仅改变了炭黑表面官能团的性质,对其孔隙结构没有明显影响。但当氧化时间达到24 h时,CBO-24的比表面积有明显下降,这可能是由炭黑粒子的过度团聚导致CBO-24表面较小的孔隙结构被掩盖。由图4(b)孔径分布曲线可以看出,4种材料表面的孔结构均以孔径为2~3.5 nm的介孔为主,其中CBO-24的介孔含量明显低于其他3种。CBO-0、CBO-6、CBO-12和CBO-24的接触角如图4(c)所示。可以看出,随着氧化时间的增加,CBO的接触角有明显的减小。证明H2O2氧化能够有效提升CBO的亲水性,使其能够更容易均匀的分布在流动电极中,并降低流动电极的泵送能量。
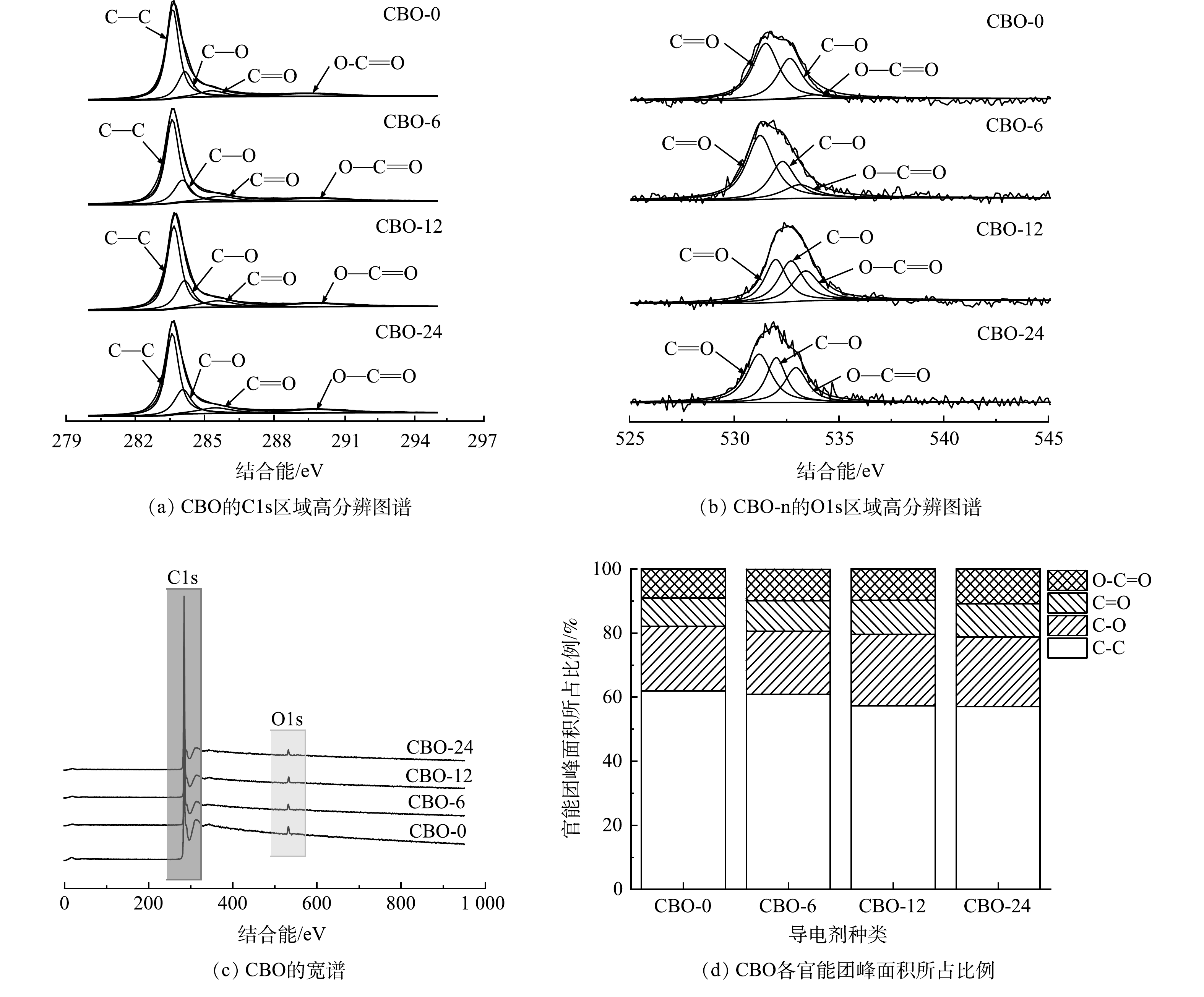
使用XPS对CBO的氧化程度和表面官能团分布情况进行分析,CBO的C1s、O1s区域高分辨谱图、宽谱和各官能团所占比例分别如图5(a)~(d)所示。由图5(a)~(b)可以看到,CBO的C1s峰均可分为C—C、C—O、C=O和O—C=O 4个峰,分别位于284.5、286.1、287.5、288.7 eV[17]。O1s峰均被分为C=O、C—O和O—C=O 3个峰,分别位于531.2、532.6和533.8 eV[18-19]。由图5(c)可以看到,CBO仅由C、O元素组成,且CBO-0、CBO-6、CBO-12和CBO-24的氧元素所占的原子百分比分别为2.3%、2.5%、2.9%和2.3%。根据CBO的C1s各分峰的面积确定了基团在CBO中所占的比例,结果如图5(d)所示。通过对比CBO的含氧官能团所占比例可以发现,随着氧化时间的升高,CBO的含氧官能团所占比例逐渐升高。其中,对比CBO-0和CBO-6可以发现,CBO-6的C=O、O—C=O基团的占比均有升高,但C—O基团的占比有所下降,证明在0~6 h内H2O2只为CB引入了少量的O元素,并将CB本来存在的C—O基团氧化为C=O和O—C=O[20-21]。当氧化时间达到12 h时,CBO-12的C—O和C=O基团所占比例明显升高,O—C=O基团含量没有明显变化,证明在6~12 h内H2O2在CB表面引入了大量C—O、C=O,C—O和C=O被氧化成O—C=O的反应所占比例很小。继续氧化至24 h,CBO-24的O—C=O基团所占比例有明显提高,且氧元素含量对比CBO-12有明显下降,这可能是由于随着氧化时间的增加,CB表面的C—O和C=O基团被继续氧化成O—C=O,且有大量含氧官能团由于H2O2的过度氧化而从CBO-24表面脱落[14, 22]。
-
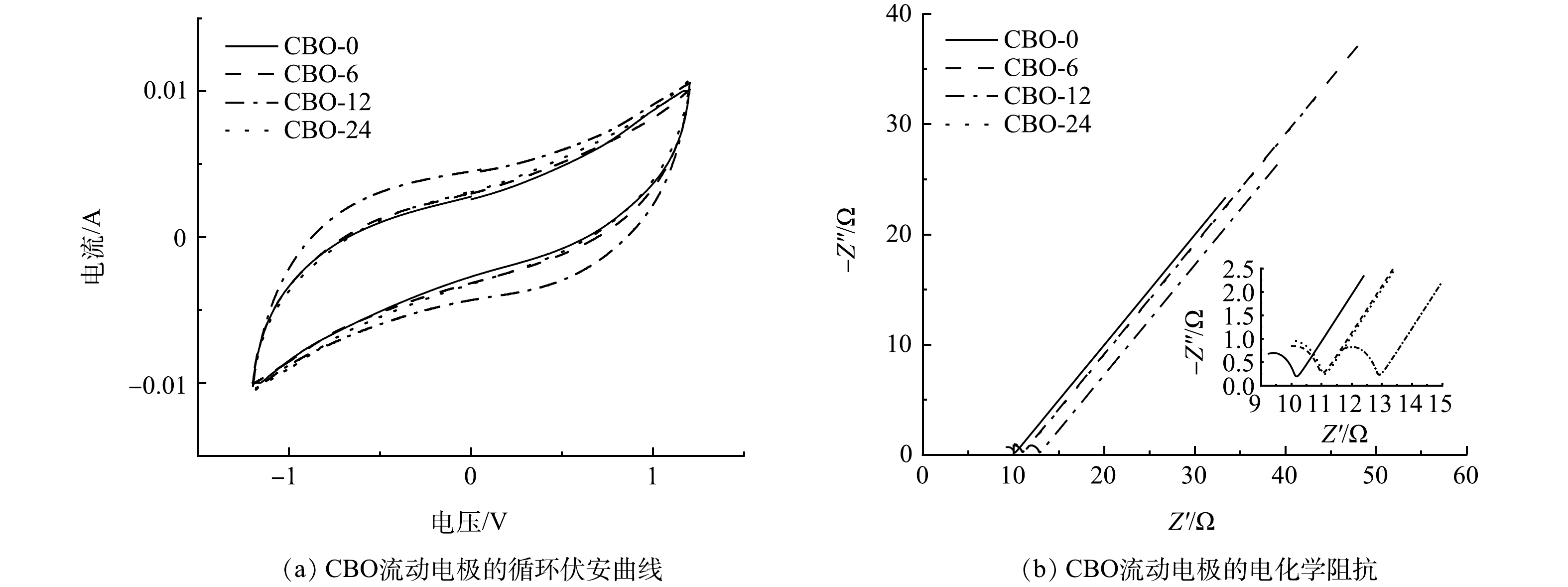
由图6(a)可以看出,由CBO配置的流动电极的CV曲线均没有出现氧化还原峰,表明其具有良好的双电层电容特性。根据式(5)计算得出,CBO-0、CBO-6、CBO-12和CBO-24的比电容分别为41.64、44.34、61.95和45.41 F∙g−1。其中,添加CBO-12的流动电极电容量有明显升高,结合图5(a)和图5(d)中CBO-0和CBO-12的C1s分峰谱和各含氧官能团在其中的占比可以看出,CB表面的C—O和C=O官能团能够有效的提高流动电极的比电容[15];进一步对比CBO-0、CBO-6和CBO-24可以发现,O—C=O对流动电极电容的提升十分有限。
CBO的EIS曲线如图6(b)所示。曲线均由高频区的半圆和低频区的直线组成,其中高频半圆的直径代表电荷转移电阻(Rct),低频斜线的斜率对应于warburg电阻(Zw)引起的离子在电解质中的扩散阻力[23]。将EIS曲线利用等效电路拟合后得出CBO-0、CBO-6、CBO-12和CBO-24的Rct分别为1.364、1.648、1.803和1.887 Ω。结合图5(b)的结果可以发现,含氧官能团所占比例的升高会导致流动电极电阻的增大。值得注意的是,虽然CB和CBO-24氧元素所占的原子百分比相似,但CBO-24的电阻明显高于CBO-0,这可能是因为CBO-24表面大量的O—C=O基团影响了CB的电子传递能力从而导致阻抗的增加[24]。
-
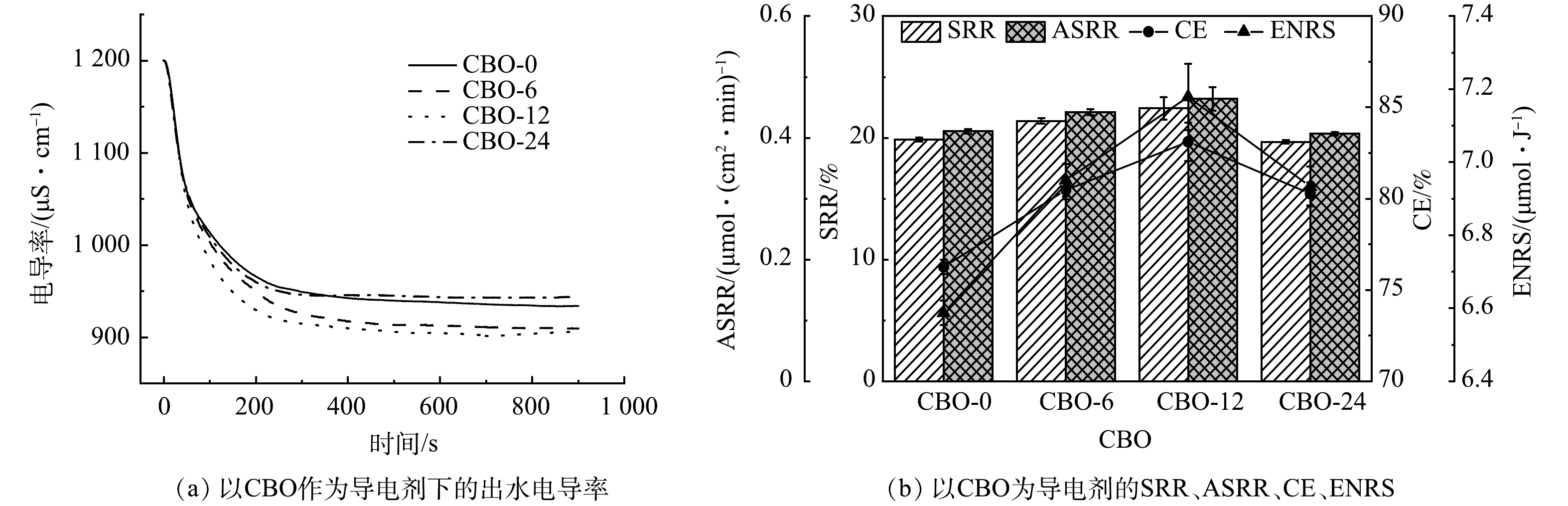
不同导电剂电极的脱盐性能如图7所示。CBO-12流动电极的脱盐性能最优,出水电导率在900 μS·cm−1左右时达到稳定,CE和ENRS分别可达到83.12%和7.18 μmol·J−1。值得注意的是,CBO-6的SRR和出水电导率与CBO-12基本一致,SRR仅相差约1%,但其CE却明显低于CBO-12。这可能是由于CBO-12表面含氧官能团所占比例的升高使其本身也具有了一定的吸附能力[25-26],为其流动电极提供了更大的电容和更多的吸附位点,使流动电极具有更优异的脱盐性能。
由图7(b)可以看出,随着CB氧化时间的增加,装置的CE和ENRS显著增加,但SRR和ASRR提升不明显,QUN等[27]对碳纸亲水性改善后电化学性能也有类似的表现。由图4(a)可以看到,由于CBO表面的孔隙结构较少,导致其吸附能力在流动电极中占比较小,导致CBO流动电极的SRR与ASRR没有明显变化。由图6(b)可以看到,随着氧化时间的升高,CBO流动电极的电荷转移电阻也会升高,这就使得单位时间内通过的电荷数量减少。但CBO亲水性的增加使其在流动电极中分散的更加均匀,远离离子交换膜的AC的双电层电容能够被充分运用,提高了电荷的利用率,从而提高了CBO流动电极的CE和ENRS[11]。值得注意的是,当氧化时间达到24 h,CBO-24流动电极的脱盐能力有明显降低,出水电导率在945 μS∙cm−1左右时就达到稳定,这是由于CBO-24被过度氧化后含氧官能团的流失使其吸附能力下降,且大量的O—C=O基团所导致的电子转移能力的下降也使得部分AC的吸附能力受到限制。对比CBO-0和CBO-24流动电极的脱盐性能可以发现,CBO-24流动电极在电阻远大于CBO-0的情况下仍能保持与CBO-0流动电极相近的SRR,这是由于CBO-24优异的亲水性使得流动电极中形成有效导电网络,使其在低电压下仍能保持较高的吸附容量。综上所述,由CBO-12作为导电剂的流动电极的脱盐性能最好,后续实验均以CBO-12作为导电剂进行。
-
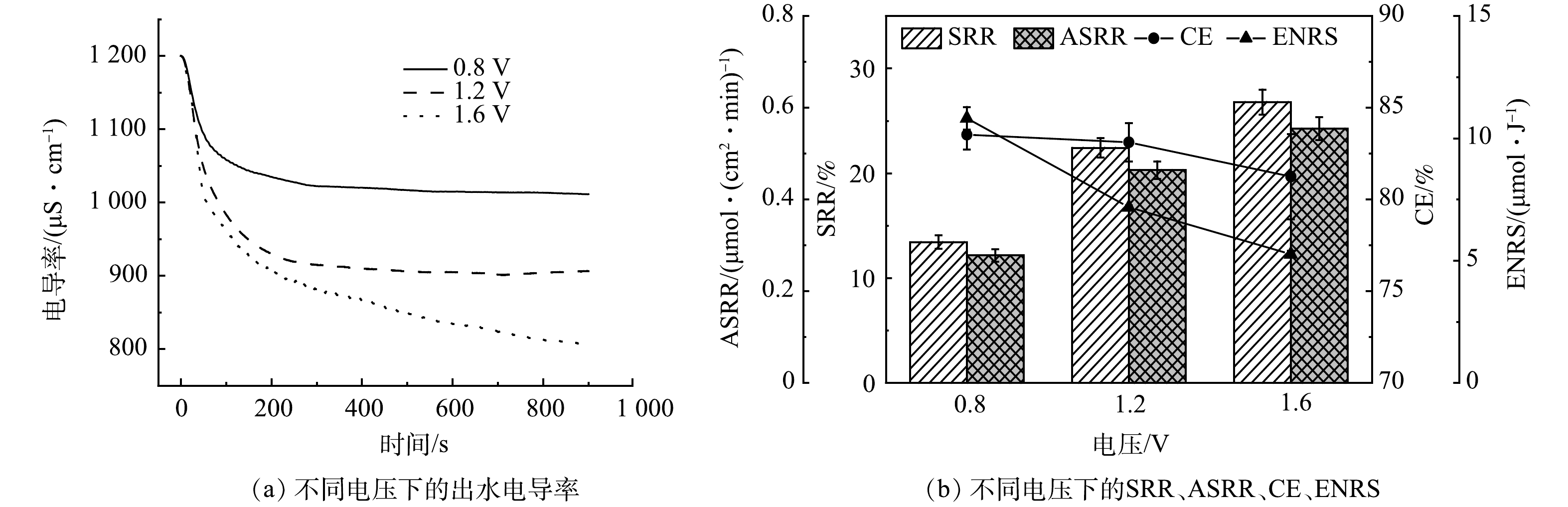
由图8可以看出,随着电压的升高,SRR有明显的提升,电压为1.6 V时的SRR是0.8 V的1.98倍(26.8%)。这是因为随着电压的升高,两电极间的电场力增强,离子迁移驱动力增加。但是,高电压也导致了CE和ENRS的迅速下降,施加电压为1.6 V时,其ENRS为5.26 μmol·J−1,仅是0.8 V时的48.6%。这可能是由于高电压下过快的电子转移无法全部用于去除盐离子所致,当电压高于水解电压1.23 V时装置中会引发法拉第效应,导致进水和流动电极中的部分水分子被电解为O2和H2,影响装置的脱盐效率。综合比较,1.2 V为最佳的加电电压。
-
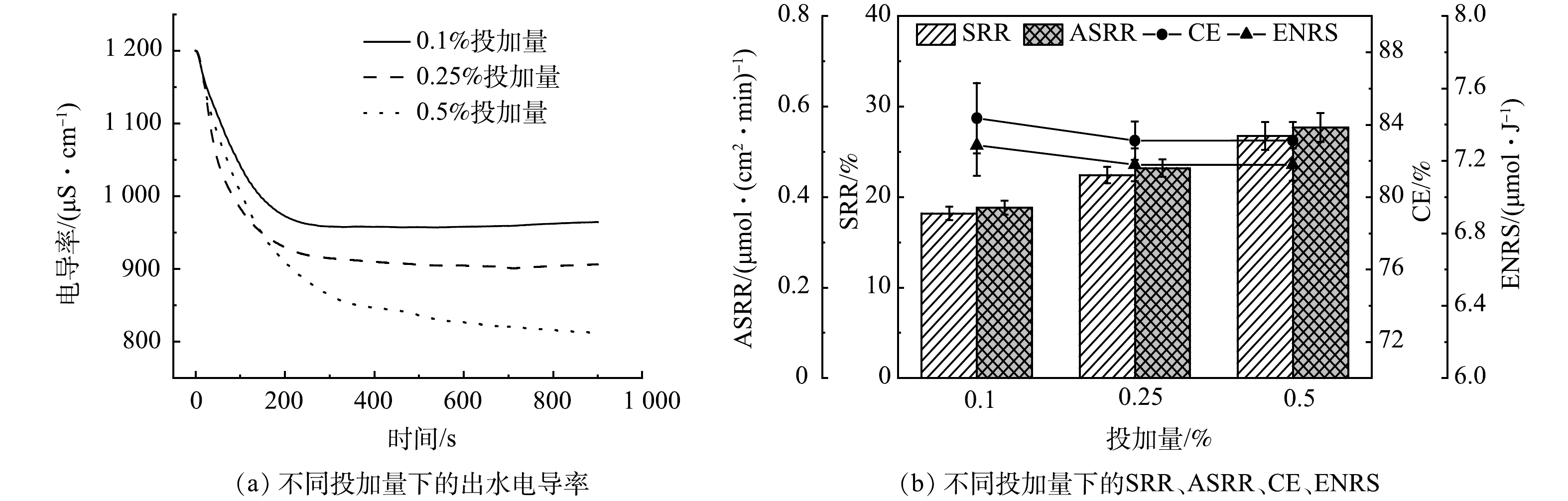
由图9可以看出,增加CBO-12的投加量能够有效的提升装置的SRR,当投加量从0.1%增加到0.5%时,SRR则从18.2%增加到26.8%,其原因主要是CBO-12本身提供了部分吸附位点。此外,由图9(b)还可以看出,随着投加量的升高,CE略有降低。这可能是因为提高CBO-12的投加量增加了电荷作用在CBO-12上的比例。但由于CBO-12的比表面积较低,导致其对离子的吸附能力和对电荷的利用率均远低于AC,从而导致CE的降低。综上所述,CBO-12最佳投加量为0.5%。值得注意的是,随着导电剂投加量的升高,流动电极的黏度会逐渐升高,增加泵送能量的同时容易引发电极在集电体流道堵塞。
-
1)氧化改性后的CBO的疏水性得到明显改善,并且其表面的含氧官能团在提高流动电极电容量的同时也使其本身也具备一定的吸附能力,但过度氧化则会导致其电荷转移电阻的升高和脱盐性能的下降。
2)氧化时间为12 h的CBO-12作为导电剂时的脱盐效果最好,电极电容量可达61.95 F∙g−1,脱盐速率可达0.46 μmol·(cm2·min)−1。
3)综合考虑流动电极脱盐效果及能耗,CBO-12流动电极的投加量为0.5%、运行电压为1.2 V时的脱盐性能最好。其脱盐率、电荷效率和单位能量脱盐量分别为26.8%、83.1%和7.18 μmol·J−1。
改性炭黑导电剂强化流动电容去离子脱盐性能的机制
Enhancement mechanism of desalination performance of the flow electrode capacitive deionization by modified carbon black conductive agent
-
摘要: 流动电极电容去离子是一种可连续运行、低能耗的新型电化学脱盐技术。在流动电极中添加导电剂是提升其脱盐性能的常用手段,但常用的导电剂如炭黑 (carbon black CB)、碳纳米管 (carbon nanotubes CNT)等的强疏水性会影响流动电极的电子传递效率和流动性,因此,提高导电剂亲水性并充分利用其吸附能力是提高流动电极脱盐性能的关键。本研究利用H2O2对炭黑进行氧化改性,通过调节氧化时间,制备了系列具有不同亲水性、表面含氧官能团的炭黑改性材料(CBO),并对其进行了详细的物理和电化学特性表征。在此基础上进一步探究了系列炭黑材料作为导电剂对装置脱盐效果的影响规律,同时优化了系统运行参数。结果表明,由H2O2氧化12 h所制备的炭黑材料作为导电剂时,装置具有较好的脱盐性能,在CBO-12投加量为0.25%、施加电压为1.2 V时,装置的平均除盐速率和电荷效率分别为0.464 μmol∙(cm2∙min)−1和83.1%。进一步表征发现,优异的脱盐性能归因于适当的氧化改性,可有效改善CB的疏水性,并在其表面引入大量含氧官能团,在改善流动电极电荷传递能力的同时,还能充分利用改性CB的吸附性能。Abstract: Flow electrode capacitive deionization is a new electrochemical desalination technology which can run continuously and has low energy consumption. Adding a conductive agent to the flow electrode is an effective method to improve the desalination performance of the flow electrode. However, the strong hydrophobicity of commonly used conductive agents (such as carbon black CB, carbon nanotubes CNT) will affect the electron transfer efficiency and the fluidity of the flow electrode. Therefore, how to increase the hydrophilicity of the conductive agent and make full use of its adsorption capacity is the key to improve the desalination performance of the flow electrode. In this study, a series of carbon black modified materials (CBO) with different hydrophilic properties and surface oxygen-containing functional groups were prepared by adjusting the oxidation time with H2O2 oxidation, and the detailed physical and electrochemical properties were characterized. On this basis, the effect of series of carbon black materials as conductive agent on the desalination effect of the device was further explored, and the operating parameters of the system were optimized. The experimental results showed that the device had a good performance when the carbon black material prepared by H2O2 oxidation for 12 hours was used as the conductive agent. When the dosage of CBO-12 was 0.25% and the applied voltage was 1.2 V, the average desalination rate and charge efficiency of the device could reach 0.464 μmol∙(cm2∙min)−1 and 83.1%, respectively. Further characterization showed that the excellent desalination performance was due to proper oxidation modification, which effectively changed the hydrophobicity of CB, and introduced a large number of oxygen-containing functional groups on its surface, which not only improved the charge transfer ability of the flow electrode, but also made full use of the adsorption performance of modified CB.
-

-
-
[1] JONGMOON C, PEMA D, HO K S, et al. Applications of capacitive deionization: Desalination, softening, selective removal, and energy efficiency[J]. Desalination, 2019, 449: 118-130. doi: 10.1016/j.desal.2018.10.013 [2] DOMENICO C, VINCENZO F, ANDREA G. A review of the water desalination technologies[J]. Applied Sciences 2021, 11(2): 2-36. [3] WANG J, SHI Z L, FANG J, et al. The optimized flow-electrode capacitive deionization (FCDI) performance by ZIF-8 derived nanoporous carbon polyhedron[J]. Separation and Purification Technology 2022, 281: 119345. [4] 吴擎昊, 马秀梅, 卢善富, 等. 利用活性碳毡构建流通式电容去离子器件及其电容脱盐性能研究[J]. 环境科学学报, 2018, 38(4): 1509-1513. [5] 刘洁, 王晓菊, 沈格, 等. 活性炭与炭黑混合电极的脱盐性能及相关工艺参数的优化[J]. 环境工程学报, 2022, 16(3): 875-885. doi: 10.12030/j.cjee.202111108 [6] WU Q H, LIANG D W, LU S F, et al. Advances and perspectives in integrated membrane capacitive deionization for water desalination[J]. Desalination 2022, 542: 116043. [7] ZHANG C Y, MA J X, WU L, et al. Flow electrode capacitive deionization (FCDI): Recent developments, environmental applications, and future perspectives[J]. Environmental Science & Technology 2021, 55(8): 4243-4267. [8] ABDULLAH A, ABDULRAHMAN A, KHALED A, et al. Surface-treated carbon black for durable, efficient, continuous flow electrode capacitive deionization[J]. Separation and Purification Technology 2023, 313: 123444. [9] CAI Y M, ZHAO X T, WANG Y, et al. Enhanced desalination performance utilizing sulfonated carbon nanotube in the flow-electrode capacitive deionization process[J]. Separation and Purification Technology 2020, 237: 116381. [10] AINOA M S, JOSE A P, MARIA I F, et al. Adsorption of Direct Blue 78 Using Chitosan and Cyclodextrins as Adsorbents[J]. Polymers 2019, 11(6): 2-18. [11] LIANG P, SUN, X L, BIAN, Y H, et al. Optimized desalination performance of high voltage flow-electrode capacitive deionization by adding carbon black in flow-electrode[J]. Desalination 2017, 420: 63-69. [12] CHO Y, YOO C Y, LEE S W, et al. Flow-electrode capacitive deionization with highly enhanced salt removal performance utilizing high-aspect ratio functionalized carbon nanotubes[J]. Water Research 2019, 151: 252-259. [13] BILEN A, PUSHPENDRA S, DEVON A E, et al. Percolation characteristics of conductive additives for capacitive flowable (semi-solid) electrodes[J]. ACS Applied Materials & Interfaces 2020, 12(5): 5866-5875. [14] FAN L Z, QIAO S Y, SONG W L. Effects of the functional groups on the electrochemical properties of ordered porous carbon for supercapacitors[J]. Electrochimica Acta 2013, 105: 299-304. [15] LI X R, JIANG Y H, WANG P Z, et al. Effect of the oxygen functional groups of activated carbon on its electrochemical performance for supercapacitors[J]. New Carbon Materials 2020, 35(3): 232-243. [16] MA J J, ZHANG C Y, YANG F, et al. Carbon black flow electrode enhanced electrochemical desalination using single-cycle operation[J]. Environmental Science & Technology 2019, 54(2): 1177-1185. [17] KUNDU S, WANG Y M, XIA W, et al. Thermal stability and reducibility of oxygen-containing functional groups on multiwalled carbon nanotube surfaces: A quantitative high-resolution XPS and TPD/ TPR study[J]. Journal of Physical Chemistry C, 2008, 112: 16869-16878. doi: 10.1021/jp804413a [18] OH H J, LEE J H, AHN H J, et al. Nanoporous activated carbon cloth for capacitive deionization of aqueous solution[J]. Thin Solid Films 2006, 515(1): 220-225. [19] YAO F B, YANG Q, YAN M, et al. Synergistic adsorption and electrocatalytic reduction of bromate by Pd/N-doped loofah sponge-derived biochar electrode[J]. Journal of Hazardous Materials 2020, 386 121651. [20] CHENG Y, HAO Z, HAO C, et al. A review of modification of carbon electrode material in capacitive deionization[J]. RSC Advances 2019, 9(42): 24401-24419. [21] HUANG W, ZHANG Y M, BAO S X, et al. Desalination by capacitive deionization process using nitric acid-modified activated carbon as the electrodes[J]. Desalination 2014, 340: 67-72. [22] MYKOLA S, DENISA H J, GAO Q L, et al. Surface functional groups of carbons and the effects of their chemical character, density and accessibility to ions on electrochemical performance[J]. Carbon 2008, 46(11): 1475-1488. [23] NIE C Y, PAN L K, LIU Y, et al. Electrophoretic deposition of carbon nanotubes–polyacrylic acid composite film electrode for capacitive deionization[J]. Electrochimica Acta 2012, 66: 106-109. [24] 谷峪. 多孔碳电极材料的常温氧化改性及其超级电容性能研究[D]. 秦皇岛: 燕山大学, 2022. [25] 刘奇杰, 吴珍珍, 江鸿雁, 等. 碳材料改性提高吸附性能的研究进展[J]. 广州化工, 2022, 50(8): 40-41. doi: 10.3969/j.issn.1001-9677.2022.08.013 [26] KELSEY B H, MARTA C H, KEVIN M C, et al. Effect of oxidation of carbon material on suspension electrodes for flow electrode capacitive deionization[J]. Environmental Science & Technology 2015, 49(5): 3040-3047. [27] LUO Q, CHENG Z Y, QIU Y F, et al. Effect of surface hydrophilicity on the supercapacitive performance of carbon paper[J]. Ionics 2017, 23(7): 1915-1920. -



 下载:
下载: