-
抗生素在人类的社会进步与经济发展中发挥了巨大的作用,被广泛的应用于医疗、畜牧、水产养殖和食品加工等行业. 然而,所使用的抗生素不能被机体完全吸收,有大于90%使用的抗生素会通过人类和动物的尿液、粪便及其他代谢途径等排放到环境中,导致抗生素在环境系统内形成持续输入与持久存在的状态,严重威胁着人类的健康和生态环境的稳定[1-4]. 由于抗生素的过量使用以及随意排放,对水环境造成了严重的污染. 因此必须研究开发一种经济有效的处理抗生素废水的方法.
目前抗生素废水的处理方法,主要为物理法、化学法和生物法. 物理法和生物法存在一些设备复杂、技术难、污染大以及费用高等问题. 光催化法是指在光照条件下,光催化剂产生的空穴和自由基与污染物作用从而去除污染物的一种方法[5-6],是目前比较热门的一种高级氧化技术. 这种高级氧化技术具有氧化能力强、反应速率快且无污染、时间少和经济成本低等优势. 该法通过特定的光使催化剂产生活性自由基团等,可氧化分解废水中的抗生素并使其最终转变为CO2和H2O等. 但是光催化材料存在着一些普遍问题,如光生载流子复合率高、量子效率低、太阳光利用率低、带隙能高不易激发以及降解反应后光催化材料不易回收,这严重限制了光催化技术的实际应用. 而无机二维纳米片具有分子或几个原子厚度以及良好的二维平面结构,是最薄的新型纳米功能材料之一[7]. 过渡金属氧化物的二维纳米片结构,一般都具有高的导电率和光响应[8]、超高的活性位点暴露比例(接近100%)[9]、大的比表面积. 其能带结构还可通过改变片层大小、厚度来调节[10]. 这些特征预示着二维纳米片很可能是一类新的、理想的光催化材料[11-12]. 当纳米片厚度小到分子或原子级别时,光生电子和空穴到达固/液或固/气界面所须迁移的距离被最大限度地减少,从而降低了电子-空穴复合的可能性. 因此,设计合成超薄二维纳米结构的光催化材料是解决光催化效率低的一个有效方法.
硫铟锌(ZnIn2S4)作为典型的阳离子合金半导体,具有对应于可见光吸收(带隙约为2.27 eV[13]). 稳定的化学性质,使之成为一种生态友好、可见光驱动的光催化剂. 然而,ZnIn2S4的光催化活性仍然不能满足更广泛应用的要求,限制其进一步适用性的基本问题之一是激发态的光诱导电子-空穴对不稳定使得在其表面处或附近容易重新结合,导致相对低的光催化效率. 因此,有效促进光生电荷载体的分离和转移,延长其寿命,是提高其光催化效率的关键. Lei等[14]首次报道用水热法合成出了在可见光下具有催化活性的ZnIn2S4;但因ZnIn2S4光生电子-空穴不稳定易复合,活性有所限制. 为了提高ZIS的光催化活性,有学者通过催化剂的复合、元素掺杂等改进手段对其进行改性[15-16]. Huang等[17]通过两步水热法合成了MoS2/ZnIn2S4复合材料,材料的比表面积增大,载流子分离效率增加,催化活性位点增多,从而表现出优异的析氢速率. 因此,将氧掺杂改性ZnIn2S4,调控ZnIn2S4表面结构,提高ZnIn2S4光生载流子的分离效率,进而提高其光催化性能. 黑磷(BP)作为一种新型的二维(2D)层状材料,自2014年首次报道场效应晶体管[11]以来,得到越来越多地关注. 由于其独特的层状结构、高电荷迁移率、0.3—2.0 eV的可调层间带隙、在UV-IR有广泛的太阳光吸收以及大比表面积和高暴露表面原子比[18],得到了越来越多的关注. 近年来,BP纳米材料作为助催化剂在光电催化、产氢和能量转换等领域被大量报道[19-20]. 由于ZnIn2S4与BP的能带匹配[21-22],研究层状ZnIn2S4与BP纳米片耦合作为降解抗生素光催化剂具有重要意义. 基于ZnIn2S4和BP的复合光催化剂的制备有望为太阳能的利用和光催化降解抗生素提供新的思路,本文通过两步水热法将氧掺杂的ZnIn2S4与二维超薄BP纳米片复合,以降解四环素为模型反应,探究不同BP复合量对光催化性能的影响,并研究了四环素的降解路径及降解机理.
-
N-甲基吡咯烷酮(NMP,C5H9NO,上海麦克林生化科技有限公司);黑磷(BP,南京先丰纳米材料科技有限公司);氢氧化钠(NaOH,上海麦克林生化科技有限公司);氯化锌(ZnCl2,上海麦克林生化科技有限公司);氯化铟(InCl3·4H2O,上海麦克林生化科技有限公司)、硫代乙酰胺(TAA,C2H5NS,上海麦克林生化科技有限公司);聚乙烯吡咯烷酮(PVP,上海麦克林生化科技有限公司);溴化钾(KBr,阿拉丁化学试剂有限公司)、乙醇(EtOH,C2H6O,阿拉丁化学试剂有限公司);四环素(TC,C22H24N2O8,上海麦克林生化科技有限公司). 所有的化学品都是分析级,整个实验所用的水都是去离子水.
-
Varian Cary 500分光光度计(UV-2600),美国;X射线衍射光谱仪(XRD, D8 Advance, Bruker Corp),德国布鲁克公司;Micromeritics trist 2030比表面积分析仪(BET),日本电子;X射线电子能谱,Kratis英国;扫描电镜(SEM, S-3400 II, Hitachi, Japan)、透射电镜(TEM, JEM-200 CX)和高分辨透射电镜(HR-TEM, Tecnai G2 F20 S),美国;电子顺磁共振波谱仪(EPR, E 500),德国Bruker;X射线光电子能谱分析(XPS, Escalab 250 XI),美国;总有机碳分析仪(German Elemental Vario TOC),德国;傅立叶变换超分辨液质联用仪(HPLC-MS),美国WATERS公司;电化学工作站(辰华Chi660E);离心机,上海安亭科学仪器厂;氙灯,北京中教金源科技有限公司;真空干燥箱,上海一恒科学仪器有限公司;电子天平,上海民桥精密科学仪器有限公司.
-
BP纳米片是通过将BP块状晶体分散于NMP试剂进行超声得到的[23]. 将50 mg的块状BP加入到50 mL的NMP溶液中,在冰水浴密封条件下用超声探针进行超声分散9 h,之后,在转速为10000 r·min−1离心机中离心5 min,并收集上清液,得到BP纳米片在NMP中的分散液.
-
将0.4 mmol ZnCl2、0.8 mmol InCl3·4H2O溶解在30 mL乙醇水溶液中,搅拌10 min,将3.2 mmol硫代乙酰胺(TAA)溶解在上述溶液中并在常温25 ℃下剧烈搅拌10 min,再称取0.2 g的PVP加入上述溶液,搅拌10 min,最后,将上述混合物溶液转移到50 mL聚四氟乙烯衬里的不锈钢高压釜中加热至180 ℃并保持24 h[24],冷却至室温后,通过离心收集样品,使用去离子水和乙醇洗涤数次,并在60 ℃的干燥烘箱中干燥10 h,研磨后得到O-ZIS样品.
-
通过静电吸引作用将BP纳米片与O-ZIS进行复合,将O-ZIS样品分别与不同体积(50、100、200、400 mL)的BP分散液混合在去离子水的密封容器中,抽真空,搅拌12 h,离心洗涤后,60 ℃干燥12 h,得到不同BP复合量的O-ZIS/BP样品. 按照BP理论复合量命名样品名称为O-ZIS、O-ZIS/0.09% BP、O-ZIS/0.23% BP、O-ZIS/0.33% BP和O-ZIS/0.5% BP. 如无特殊说明,O-ZIS/BP特指O-ZIS/0.5% BP.
-
通过降解TC测试催化剂的光催化活性,室温(20±3)℃条件下,在250 mL的烧杯中将20 mg催化剂样品分散在40 mL TC(1×10−4 mol·L−1)溶液中. 首先,在黑暗中磁力搅拌悬浮液30 min,达到吸附-解吸平衡后,开氙灯(300 W,λ≥420 nm). 经过一定的时间间隔,吸取3 mL样品,经滤头(0.45 μm)过滤后,用紫外-可见分光光度计在TC的特征吸收波长357 nm处测定样品吸光度,并进而分析TC的浓度变化[25]. TC溶液的去除率μ根据式(1)进行计算.
式中:μ 为降解率,%;C 和C0 分别表示TC的降解后浓度和初始浓度,mg·L−1.
-
通过德国Bruker公司型号为E 500的电子顺磁共振波谱仪测试在光催化反应过程中产生的自由基种类. 羟基自由基·OH的测试:称取5 mg催化剂加入5 mL去离子水,超声5 min分散均匀,后加入5,5-二甲基-1-吡咯-N-氧化物(5,5-dimethyl-1-pyrroline-N-oxide,DMPO). 氙灯下照射5 min后取样,用毛细管吸取液体后用真空脂密封,放入EPR管测试自由基信号. 对于超氧自由基·O2−的测试为称取5 mg催化剂加入5 mL甲醇,超声5 min分散均匀,后加入5,5-二甲基-1-吡咯-N-氧化物(5,5-dimethyl-1-pyrroline-N-oxide,DMPO). 氙灯下照射5 min后取样,用毛细管吸取液体后用真空脂密封,放入EPR管测试自由基信号.
-
由于在光催化反应过程中,体系中会产生受光照激发的电子,所以可以使用电化学工作站评估所制备样品的光电化学性能. 采用的电化学工作站型号为CHI660E. 具体条件为:在含有0.2 mol·L−1 Na2SO4电解质溶液的石英电解池中采用传统的三电极体系进行实验,对电极为铂(Pt)片电极,参比电极为Ag/AgCl电极,工作电极为涂有样品的FTO导电玻璃[26]. 催化剂光电流的测试条件为:光照间隔20 s,取样时长为300 s. 工作电极的制备:称取5 mg样品,加入200 μL乙醇溶液中,再滴加两滴萘酚,让催化剂粉末更好的粘在导电玻璃上,超声30 min,混合均匀后用移液枪取10 μL的混合液,滴在导电玻璃片上,自然风干后,再进行电化学测试[27].
-
为了定量四环素在催化剂作用下降解后的矿化程度,对四环素最终降解产物进行总有机碳含量分析. 待光照一定时间后,取10 mL反应溶液,经0.45 μm针式滤头过滤后,用TOC 分析仪进行分析测试. 采用下面公式得到四环素最终矿化程度:
式中,TOC0为初始四环素溶液总有机碳含量,TOCt为降解后总有机碳含量,t为可见光照射时间,min.
-
为了探究光催化反应过程中四环素分子降解的路径,在降解完成后收集降解产物,用色谱纯级乙腈稀释产物至大约0.005 mg·L−1,利用美国WATERS公司的液相-高分辨质谱联用仪进行分析,采用RESTEK Ultra C18色谱柱(4.6 mm× 250 mm,粒径5 µm),二极管阵列检测器;流动相由30%水(含0.5%磷酸)(A)和70%甲醇(B)组成,流速为0.80 mL·min−1;进样量为20 µL,柱温为30 ℃. 根据质谱峰的荷质比最终确定反应过程中产生的中间产物分子.
-
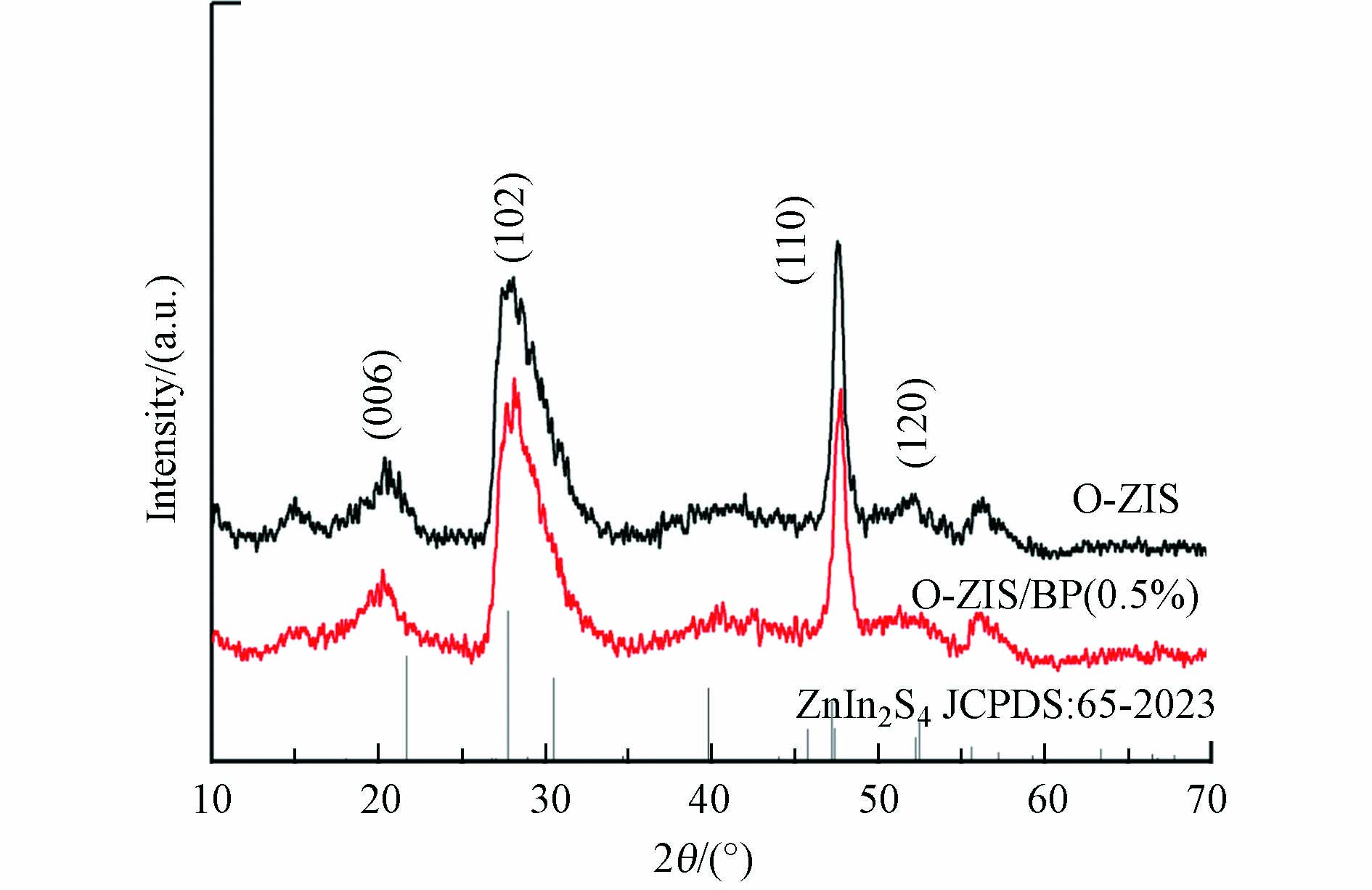
通过XRD对制备的O-ZIS和O-ZIS/BP样品进行晶体结构表征. 如图1所示,对比ZnIn2S4标准卡片,可以看出O-ZIS样品的特征衍射峰向低角度偏移,这可能是由于氧掺杂导致ZIS产生晶格畸变[12];还可得知合成的O-ZIS样品为六方晶相ZnIn2S4,2θ为27.7°和47.2°处分别对应于ZnIn2S4(102)和(110)晶面. 对比O-ZIS/BP与O-ZIS样品的XRD谱图,可以看到二者的出峰位置和峰高没有明显的变化,表明BP的复合没有改变O-ZIS的晶体结构. 谱图中没有BP的特征峰出现,可能是由于BP含量少,未达到检测限.
-
采用扫描电镜(SEM)和透射电镜(TEM)观察样品的形貌结构. 从低倍SEM(图2a)图中可以看出,超声分散后的BP呈现纳米片状形貌;所制备的O-ZIS(图2b)纳米片是呈类花状结构,可以清楚地观察到它是由很多个纳米片聚合组装成的. 图2c为复合样品的SEM图,可以看到BP纳米片均匀地附着在O-ZIS纳米微球表面. 通过TEM(图2d)可以清楚地看到O-ZIS/BP复合样品超薄层状纳米片结构;从高分辨透射电镜(HRTEM)(图2e)中可以清晰地看到O-ZIS和BP纳米片之间紧密的界面接触,还可以清晰地观察到晶格条纹,晶格间距为0.33 nm对应于O-ZIS的(102)晶面,0.37 nm的晶格间距则对应于BP纳米片的(111)晶面. 这一结果也证实了O-ZIS纳米薄片与BP纳米片的成功复合.
-
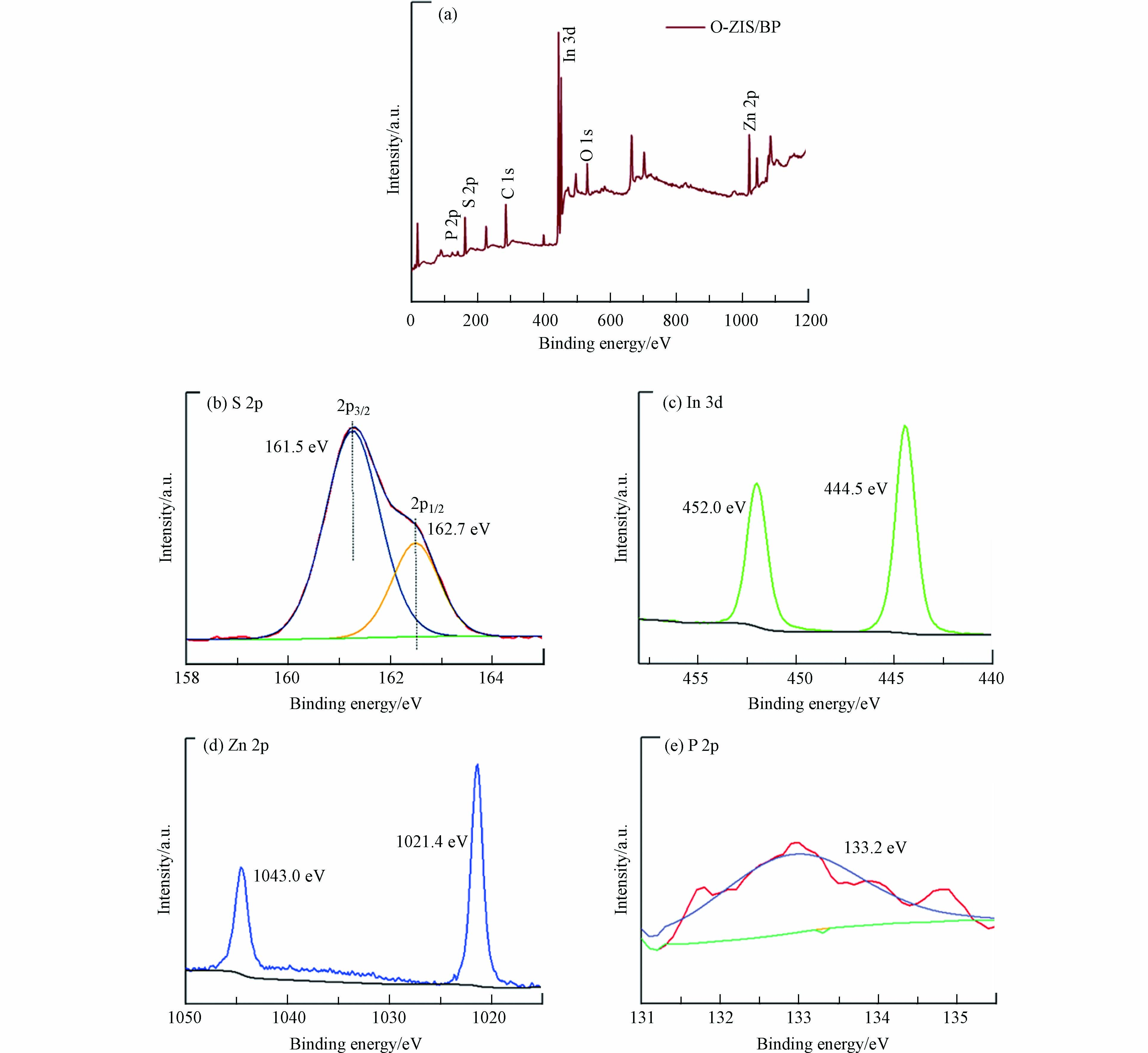
用X射线光电子能谱(XPS)分析了O-ZIS/BP样品的表面组成、化学状态以及元素之间的相互作用. XPS全谱图(图3a)显示,样品含有Zn、In、O、S、P元素,表明BP与O-ZIS的成功复合. 从S 2p 的高分辨XPS谱图(图3b)可见,在结合能为161.5 eV和162.7 eV处的两个特征峰分别归属于S 2p3/2 和S 2p1/2,是S2-离子的特征峰[28];In 3d高分辨XPS谱图(图3c)显示位于444.5 eV和452.0 eV两个特征峰,分别对应于In 3d5/2、In 3d3/2的结合能;图3d为Zn 2p高分辨XPS谱图,特征峰位于1021.4 eV和1043.0 eV,分别属于Zn 2p3/2和Zn 2p1/2结合能,为正二价锌[29];样品的P 2p高分辨能谱(图3e)呈现出结合能133.2 eV的特征峰,通过分析得出特征峰归属于P-O键,可能是BP中的P与O-ZIS中的O成键[30]. XPS表征结果证明BP与O-ZIS的复合是化学键作用,而非简单的机械混合.
-
通过在可见光(λ≥420 nm)照射下降解TC考察了样品的光催化性能. 在没有光照的条件下,先进行了30 min暗吸附,让体系达到吸附平衡. 如图4(a)所示,TC只是在光照而无催化剂条件下是不会发生自分解的,必须要在催化剂和光照共同作用下,才能实现TC的降解;相较于O-ZIS样品,O-ZIS/BP样品对TC的吸附能力略有增加,这可能是由于O-ZIS样品在复合BP后比表面积有所增加. 另外,在可见光照射下,复合材料降解TC的能力显著提高. 对比不同BP复合量的O-ZIS/BP催化剂(O-ZIS、O-ZIS/0.09% BP、O-ZIS/0.23% BP、O-ZIS/0.33% BP和O-ZIS/0.5% BP)的催化活性可以看出,BP的复合量为0.5%时,降解效果最佳,光照20 min,TC降解率达到90%,远高于空白O-ZIS样品的60%.
继续延长光照时间到40 min,TC降解率达到99%. 而继续增加BP负载量使得O-ZIS表面被覆盖,纳米片的活性位点减少,从而限制了其光催化性能. 这一结果表明BP的复合能极大提高ZIS光催化降解TC的活性.
由于TC的降解从实验方法上可以看出TC浓度仅仅是通过对特征峰值的大小来判断,但是在实际反应中,降解不是一个一步矿化的过程,反应过程中产生的自由基不断攻击四环素分子上的化学键,逐渐将其矿化成H2O和CO2,因此评价催化剂的降解能力还需要通过总有机碳的测试来判断[31]. 如图4(b)所示,降解反应30 min后整体矿化能力与其降解活性趋势一致,O-ZIS/BP催化剂具有比O-ZIS更优的矿化率,TOC去除率达到了64.5%,表现出较好的矿化能力,降低了环境安全风险.
-
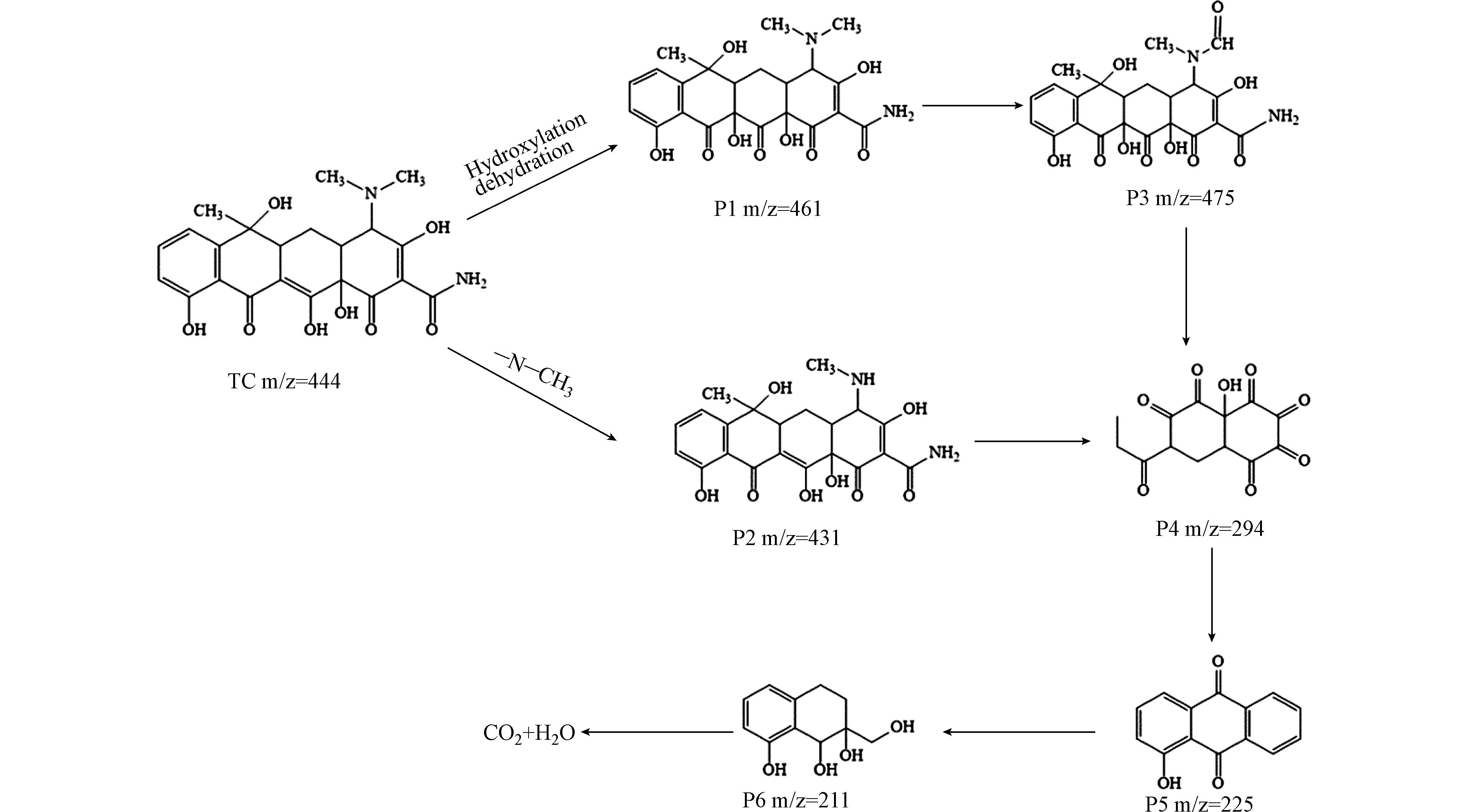
为了进一步研究O-ZIS/BP光催化降解TC的路径、中间产物以及活性物种作用的基团和键能,理解复合物光催化降解四环素的机理,本工作采用HPLC-MS分析了最终反应产物. 中间产物的降解路径图及荷质比m/z值如图5所示. 四环素具有双键、酚键和氨基基团,这些基团的电子密度较高,易受到·O2−和·OH自由基的攻击然后断开,尤其是双键. 在催反应的最初,由于N-C的键能较低,N—CH3之间的键会首先被断开,形成荷质比m/z = 431的化合物. 另一个反应路径主要是羟基化反应过程,首先是一个羟基基团攻击四环素分子,形成m/z = 461的化合物,然后羟基基团继续攻击,生成其他的分子化合物. 从图5中可以清楚的看出,在可见光照射下,四环素逐渐降解为一些分子量越来越小的中间体,如m/z为475、294、225、211的分子. 在整个降解过程中四环素的4个环被一步步打开,最终被矿化成CO2、H2O和无机离子[32].
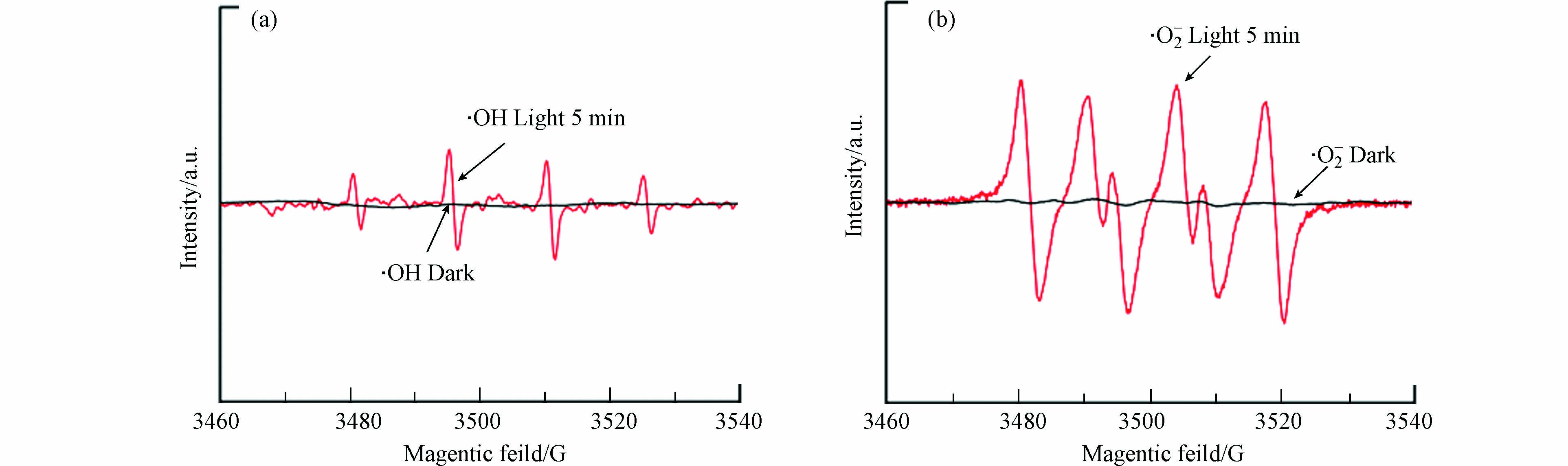
为了进一步探究O-ZIS/BP在可见光照射下反应体系中产生的活性氧物种,采用电子自旋共振(EPR)对反应体系进行了表征. 实验以DMPO为自由基捕获剂,分别对于羟基自由基(·OH)和超氧自由基(·O2−)进行测试,结果如图6所示. 黑暗条件下在催化剂的甲醇分散液中未发现明显的EPR信号,如图6(a)所示,在黑暗条件下,催化剂的水分散体系中未检测到明显的DMPO-·OH特征信号,而在光照射条件下,出现明显的 DMPO-·OH特征峰信号,表明O-ZIS/BP在光催化降解过程中产生了·OH自由基. 此外,如图6(b)所示,在光照5 min后,出现的DMPO-·O2−特征峰信号,归属于·O2−活性物质的产生,表明溶液中O2被O-ZIS/BP的光生电子还原生成·O2−自由基,并作为主要活性物种参与光催化降解反应. 因此,EPR表征结果说明O-ZIS/BP光催化过程产生了·OH和·O2−自由基,在降解TC过程中起到了主要作用.
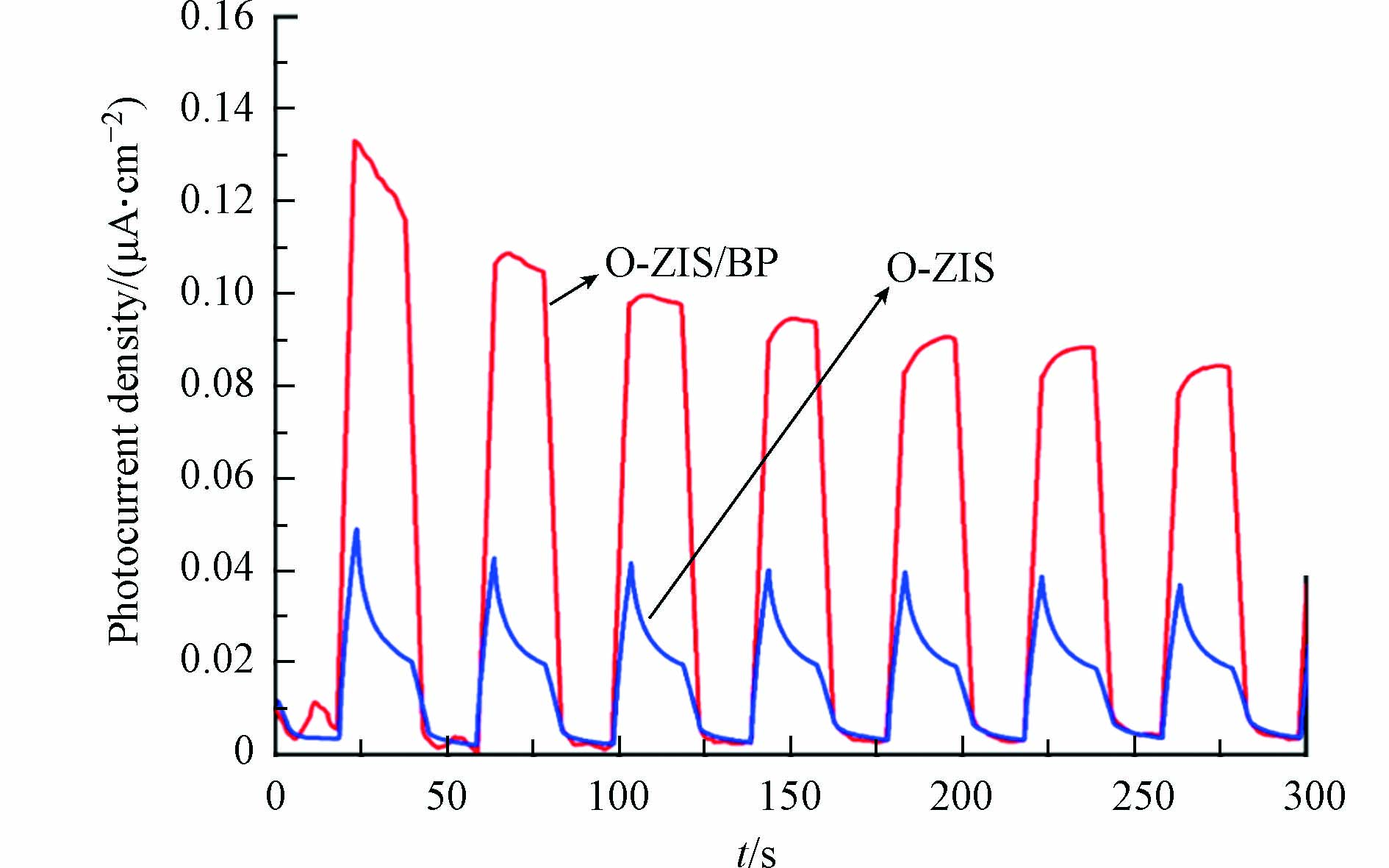
由于光催化反应过程中光会激发半导体价带上的电子,因此可以通过电化学的表征来研究光催化剂的电荷分离效率. 其中,光电流测试是评估光生载流子分离和传输效率的方法之一. 本实验通过光电流测试表征了样品光生电子的分离效率,结果如图7所示,空白O-ZIS样品的光电流响应强度较小,说明O-ZIS的电子-空穴分离效率比较低;O-ZIS/BP复合样品的光电流响应强度有明显增加,大约是O-ZIS空白样品的3倍,且经过7轮开关灯循环,其光电流强度仍能保持较高的强度,说明对O-ZIS进行BP复合可以显著提高其光生载流子的分离效率,有利于光催化性能的提高[33].
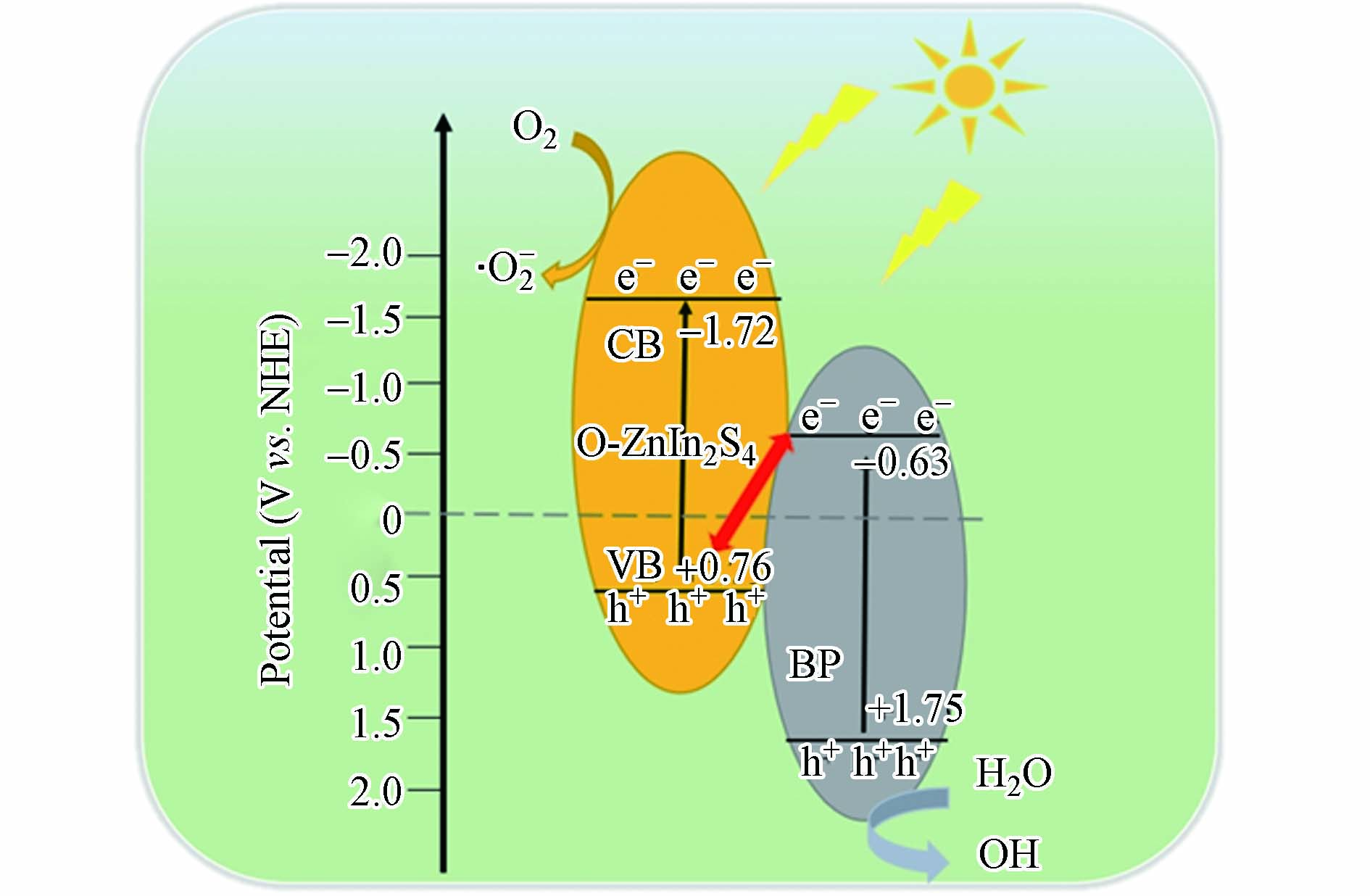
基于以上表征结果分析, O-ZIS/BP复合物提高光催化降解TC活性的可能机理,如图8所示. 前期报道过O-ZIS的导带(CB)和价带(VB)电势分别是-1.72 eV和0.76 eV[11],并利用DRS测得的样品吸收带边计算出O-ZIS的禁带宽度为2.48 eV;另外,文献报道剥离的BP纳米片的导带(CB)和价带(VB)电势分别是- 0.63 eV和1.75 eV,表明BP能很好地与O-ZIS实现能带匹配,形成异质结[34]. 因此,在可见光照射下,O-ZIS和BP中的价带电子均被激发至导带,BP导带电子与O-ZIS价带空穴通过致密的相界面复合,实现了O-ZIS和BP自身电子空穴对的有效分离,从而极大提高了其光催化活性. BP价带空穴氧化异质结表面吸附的水产生了·OH,O-ZIS导带电子还原O2产生·O2-,这些具有强氧化能力的自由基将TC氧化降解. 可以看出,O-ZIS/BP复合物光催化降解四环素(TC)反应过程如下:
-
通过水热法合成O-ZIS纳米片,再通过静电吸附作用将超声剥离的BP纳米片成功复合在O-ZIS纳米片表面,形成O-ZIS/BP复合催化剂. BP的复合没有改变O-ZIS的晶体结构,BP纳米片能与O-ZIS纳米片形成紧密的界面接触,且形成了P—O化学键. 复合样品在可见光下表现出优异的光催化降解四环素性能,BP的复合量对O-ZIS材料的光催化性能也有影响,O-ZIS/0.5%wt BP样品的降解活性最佳,反应20 min,TC降解率达到90%,矿化率达到64.5%,远高于空白O-ZIS样品. 光电流测试结果表明BP纳米片的复合能显著促进光生电子和空穴对的分离效率. ·O2−和·OH自由基在O-ZIS/BP光催化降解反应过程中起到主要作用,能实现四环素的开环分解,直至完全矿化. 该结果将为开发通过BP纳米片复合光催化剂的方法来提高光催化活性提供新的思路.
O-ZnIn2S4/BP光催化剂的制备及降解四环素研究
Preparation of O-ZnIn2S4 /BP photocatalyst and study on its degradation of tetracycline
-
摘要: 抗生素的过量使用以及随意排放,对水环境造成了严重的污染. 光催化降解技术是一种经济有效的处理抗生素废水的方法,高效光催化材料的设计合成是该技术实现实际应用的关键. 层状ZnIn2S4(ZIS)作为典型的阳离子合金半导体,不仅具有较窄的带隙,能有效吸收可见光,而且还拥有较高的平带电势和稳定的化学性质,使其成为了一种很具应用前景的可见光催化材料. 然而ZIS材料光生载流子分离效率低还无法实现实际应用. 本文通过温和的两步法将氧掺杂的ZnIn2S4(O-ZIS)和黑磷(BP)纳米片进行复合,提高ZIS的光生载流子分离效率. 结果表明,在可见光照射下,复合催化剂O-ZIS/BP能高效降解四环素(TC),在BP最佳复合量(0.5%)时,光照20 min,TC降解率达到90%,矿化率达到64.5%. BP的复合能显著增强光电流响应、电子迁移速率. ·O2−和·OH自由基在O-ZIS/BP 光催化降解TC反应过程中起到主要作用. 该研究将为开发二维纳米片复合光催化剂及其在环境修复领域的应用提供新的思路.
-
关键词:
- 光催化 /
- ZnIn2S4/BP /
- 四环素降解 /
- 纳米片
Abstract: The excessive use and random discharge of antibiotics have caused serious pollution to the water environment. Photocatalytic degradation technology is an economical and effective method to treat antibiotic wastewater. The design and synthesis of highly efficient photocatalytic materials is the key to the practical application of this technology. As a typical cationic alloy semiconductor, layered ZnIn2S4 (ZIS) not only has a narrow band gap and can effectively absorb visible light, but also has a high flat band potential and stable chemical properties, which makes layered ZIS become a visible light catalytic material with great application prospect. However, ZIS material has the disadvantage of low photogenerated carrier separation efficiency, which limits the practical application of ZIS. In this study, oxygen doped ZnIn2S4 (O-ZIS) and black phosphorus (BP) nanosheet were combined by a mild two-step method to prepare a composite photocatalyst (O-ZIS/BP). Under visible light irradiation, the highly efficient degradation of pollutant tetracycline (TC) was achieved. The recombination of BP can significantly enhance the electron migration rate in photocurrent response. When the recombination amount of BP reaches 0.5%, the best activity is achieved with TC degradation rate of 90% and TOC removal of 64.5% under 20 min of light. ·O2− and ·OH radicals play an important role in O-ZIS /BP photocatalytic TC degradation. This study will provide a new idea for the development of two-dimensional nanosheet composite photocatalyst and its application in environmental remediation.-
Key words:
- photocatalysis /
- ZnIn2S4/BP /
- tetracycline degradation /
- nanosheets.
-
抗生素在人类的社会进步与经济发展中发挥了巨大的作用,被广泛的应用于医疗、畜牧、水产养殖和食品加工等行业. 然而,所使用的抗生素不能被机体完全吸收,有大于90%使用的抗生素会通过人类和动物的尿液、粪便及其他代谢途径等排放到环境中,导致抗生素在环境系统内形成持续输入与持久存在的状态,严重威胁着人类的健康和生态环境的稳定[1-4]. 由于抗生素的过量使用以及随意排放,对水环境造成了严重的污染. 因此必须研究开发一种经济有效的处理抗生素废水的方法.
目前抗生素废水的处理方法,主要为物理法、化学法和生物法. 物理法和生物法存在一些设备复杂、技术难、污染大以及费用高等问题. 光催化法是指在光照条件下,光催化剂产生的空穴和自由基与污染物作用从而去除污染物的一种方法[5-6],是目前比较热门的一种高级氧化技术. 这种高级氧化技术具有氧化能力强、反应速率快且无污染、时间少和经济成本低等优势. 该法通过特定的光使催化剂产生活性自由基团等,可氧化分解废水中的抗生素并使其最终转变为CO2和H2O等. 但是光催化材料存在着一些普遍问题,如光生载流子复合率高、量子效率低、太阳光利用率低、带隙能高不易激发以及降解反应后光催化材料不易回收,这严重限制了光催化技术的实际应用. 而无机二维纳米片具有分子或几个原子厚度以及良好的二维平面结构,是最薄的新型纳米功能材料之一[7]. 过渡金属氧化物的二维纳米片结构,一般都具有高的导电率和光响应[8]、超高的活性位点暴露比例(接近100%)[9]、大的比表面积. 其能带结构还可通过改变片层大小、厚度来调节[10]. 这些特征预示着二维纳米片很可能是一类新的、理想的光催化材料[11-12]. 当纳米片厚度小到分子或原子级别时,光生电子和空穴到达固/液或固/气界面所须迁移的距离被最大限度地减少,从而降低了电子-空穴复合的可能性. 因此,设计合成超薄二维纳米结构的光催化材料是解决光催化效率低的一个有效方法.
硫铟锌(ZnIn2S4)作为典型的阳离子合金半导体,具有对应于可见光吸收(带隙约为2.27 eV[13]). 稳定的化学性质,使之成为一种生态友好、可见光驱动的光催化剂. 然而,ZnIn2S4的光催化活性仍然不能满足更广泛应用的要求,限制其进一步适用性的基本问题之一是激发态的光诱导电子-空穴对不稳定使得在其表面处或附近容易重新结合,导致相对低的光催化效率. 因此,有效促进光生电荷载体的分离和转移,延长其寿命,是提高其光催化效率的关键. Lei等[14]首次报道用水热法合成出了在可见光下具有催化活性的ZnIn2S4;但因ZnIn2S4光生电子-空穴不稳定易复合,活性有所限制. 为了提高ZIS的光催化活性,有学者通过催化剂的复合、元素掺杂等改进手段对其进行改性[15-16]. Huang等[17]通过两步水热法合成了MoS2/ZnIn2S4复合材料,材料的比表面积增大,载流子分离效率增加,催化活性位点增多,从而表现出优异的析氢速率. 因此,将氧掺杂改性ZnIn2S4,调控ZnIn2S4表面结构,提高ZnIn2S4光生载流子的分离效率,进而提高其光催化性能. 黑磷(BP)作为一种新型的二维(2D)层状材料,自2014年首次报道场效应晶体管[11]以来,得到越来越多地关注. 由于其独特的层状结构、高电荷迁移率、0.3—2.0 eV的可调层间带隙、在UV-IR有广泛的太阳光吸收以及大比表面积和高暴露表面原子比[18],得到了越来越多的关注. 近年来,BP纳米材料作为助催化剂在光电催化、产氢和能量转换等领域被大量报道[19-20]. 由于ZnIn2S4与BP的能带匹配[21-22],研究层状ZnIn2S4与BP纳米片耦合作为降解抗生素光催化剂具有重要意义. 基于ZnIn2S4和BP的复合光催化剂的制备有望为太阳能的利用和光催化降解抗生素提供新的思路,本文通过两步水热法将氧掺杂的ZnIn2S4与二维超薄BP纳米片复合,以降解四环素为模型反应,探究不同BP复合量对光催化性能的影响,并研究了四环素的降解路径及降解机理.
1. 实验部分(Experiment section)
1.1 实验原料
N-甲基吡咯烷酮(NMP,C5H9NO,上海麦克林生化科技有限公司);黑磷(BP,南京先丰纳米材料科技有限公司);氢氧化钠(NaOH,上海麦克林生化科技有限公司);氯化锌(ZnCl2,上海麦克林生化科技有限公司);氯化铟(InCl3·4H2O,上海麦克林生化科技有限公司)、硫代乙酰胺(TAA,C2H5NS,上海麦克林生化科技有限公司);聚乙烯吡咯烷酮(PVP,上海麦克林生化科技有限公司);溴化钾(KBr,阿拉丁化学试剂有限公司)、乙醇(EtOH,C2H6O,阿拉丁化学试剂有限公司);四环素(TC,C22H24N2O8,上海麦克林生化科技有限公司). 所有的化学品都是分析级,整个实验所用的水都是去离子水.
1.2 实验设备
Varian Cary 500分光光度计(UV-2600),美国;X射线衍射光谱仪(XRD, D8 Advance, Bruker Corp),德国布鲁克公司;Micromeritics trist 2030比表面积分析仪(BET),日本电子;X射线电子能谱,Kratis英国;扫描电镜(SEM, S-3400 II, Hitachi, Japan)、透射电镜(TEM, JEM-200 CX)和高分辨透射电镜(HR-TEM, Tecnai G2 F20 S),美国;电子顺磁共振波谱仪(EPR, E 500),德国Bruker;X射线光电子能谱分析(XPS, Escalab 250 XI),美国;总有机碳分析仪(German Elemental Vario TOC),德国;傅立叶变换超分辨液质联用仪(HPLC-MS),美国WATERS公司;电化学工作站(辰华Chi660E);离心机,上海安亭科学仪器厂;氙灯,北京中教金源科技有限公司;真空干燥箱,上海一恒科学仪器有限公司;电子天平,上海民桥精密科学仪器有限公司.
1.3 实验方法
1.3.1 BP纳米片的制备
BP纳米片是通过将BP块状晶体分散于NMP试剂进行超声得到的[23]. 将50 mg的块状BP加入到50 mL的NMP溶液中,在冰水浴密封条件下用超声探针进行超声分散9 h,之后,在转速为10000 r·min−1离心机中离心5 min,并收集上清液,得到BP纳米片在NMP中的分散液.
1.3.2 氧掺杂ZnIn2S4 (O-ZIS) 纳米片的制备
将0.4 mmol ZnCl2、0.8 mmol InCl3·4H2O溶解在30 mL乙醇水溶液中,搅拌10 min,将3.2 mmol硫代乙酰胺(TAA)溶解在上述溶液中并在常温25 ℃下剧烈搅拌10 min,再称取0.2 g的PVP加入上述溶液,搅拌10 min,最后,将上述混合物溶液转移到50 mL聚四氟乙烯衬里的不锈钢高压釜中加热至180 ℃并保持24 h[24],冷却至室温后,通过离心收集样品,使用去离子水和乙醇洗涤数次,并在60 ℃的干燥烘箱中干燥10 h,研磨后得到O-ZIS样品.
1.3.3 BP复合O-ZIS样品的制备
通过静电吸引作用将BP纳米片与O-ZIS进行复合,将O-ZIS样品分别与不同体积(50、100、200、400 mL)的BP分散液混合在去离子水的密封容器中,抽真空,搅拌12 h,离心洗涤后,60 ℃干燥12 h,得到不同BP复合量的O-ZIS/BP样品. 按照BP理论复合量命名样品名称为O-ZIS、O-ZIS/0.09% BP、O-ZIS/0.23% BP、O-ZIS/0.33% BP和O-ZIS/0.5% BP. 如无特殊说明,O-ZIS/BP特指O-ZIS/0.5% BP.
1.3.4 光催化降解实验
通过降解TC测试催化剂的光催化活性,室温(20±3)℃条件下,在250 mL的烧杯中将20 mg催化剂样品分散在40 mL TC(1×10−4 mol·L−1)溶液中. 首先,在黑暗中磁力搅拌悬浮液30 min,达到吸附-解吸平衡后,开氙灯(300 W,λ≥420 nm). 经过一定的时间间隔,吸取3 mL样品,经滤头(0.45 μm)过滤后,用紫外-可见分光光度计在TC的特征吸收波长357 nm处测定样品吸光度,并进而分析TC的浓度变化[25]. TC溶液的去除率μ根据式(1)进行计算.
μ=(1−C/C0)×100% (1) 式中:μ 为降解率,%;C 和C0 分别表示TC的降解后浓度和初始浓度,mg·L−1.
1.3.5 电子顺磁共振(EPR)实验
通过德国Bruker公司型号为E 500的电子顺磁共振波谱仪测试在光催化反应过程中产生的自由基种类. 羟基自由基·OH的测试:称取5 mg催化剂加入5 mL去离子水,超声5 min分散均匀,后加入5,5-二甲基-1-吡咯-N-氧化物(5,5-dimethyl-1-pyrroline-N-oxide,DMPO). 氙灯下照射5 min后取样,用毛细管吸取液体后用真空脂密封,放入EPR管测试自由基信号. 对于超氧自由基·O2−的测试为称取5 mg催化剂加入5 mL甲醇,超声5 min分散均匀,后加入5,5-二甲基-1-吡咯-N-氧化物(5,5-dimethyl-1-pyrroline-N-oxide,DMPO). 氙灯下照射5 min后取样,用毛细管吸取液体后用真空脂密封,放入EPR管测试自由基信号.
1.3.6 光电流测试
由于在光催化反应过程中,体系中会产生受光照激发的电子,所以可以使用电化学工作站评估所制备样品的光电化学性能. 采用的电化学工作站型号为CHI660E. 具体条件为:在含有0.2 mol·L−1 Na2SO4电解质溶液的石英电解池中采用传统的三电极体系进行实验,对电极为铂(Pt)片电极,参比电极为Ag/AgCl电极,工作电极为涂有样品的FTO导电玻璃[26]. 催化剂光电流的测试条件为:光照间隔20 s,取样时长为300 s. 工作电极的制备:称取5 mg样品,加入200 μL乙醇溶液中,再滴加两滴萘酚,让催化剂粉末更好的粘在导电玻璃上,超声30 min,混合均匀后用移液枪取10 μL的混合液,滴在导电玻璃片上,自然风干后,再进行电化学测试[27].
1.3.7 总有机碳(TOC)测试
为了定量四环素在催化剂作用下降解后的矿化程度,对四环素最终降解产物进行总有机碳含量分析. 待光照一定时间后,取10 mL反应溶液,经0.45 μm针式滤头过滤后,用TOC 分析仪进行分析测试. 采用下面公式得到四环素最终矿化程度:
TOC去除率(%)=(TOC0−TOCtTOC0)×100% (2) 式中,TOC0为初始四环素溶液总有机碳含量,TOCt为降解后总有机碳含量,t为可见光照射时间,min.
1.3.8 高分辨液-质联用(HPLC-MS)测试
为了探究光催化反应过程中四环素分子降解的路径,在降解完成后收集降解产物,用色谱纯级乙腈稀释产物至大约0.005 mg·L−1,利用美国WATERS公司的液相-高分辨质谱联用仪进行分析,采用RESTEK Ultra C18色谱柱(4.6 mm× 250 mm,粒径5 µm),二极管阵列检测器;流动相由30%水(含0.5%磷酸)(A)和70%甲醇(B)组成,流速为0.80 mL·min−1;进样量为20 µL,柱温为30 ℃. 根据质谱峰的荷质比最终确定反应过程中产生的中间产物分子.
2. 结果与讨论(Results and discussion)
2.1 O-ZIS/BP样品的XRD图谱
通过XRD对制备的O-ZIS和O-ZIS/BP样品进行晶体结构表征. 如图1所示,对比ZnIn2S4标准卡片,可以看出O-ZIS样品的特征衍射峰向低角度偏移,这可能是由于氧掺杂导致ZIS产生晶格畸变[12];还可得知合成的O-ZIS样品为六方晶相ZnIn2S4,2θ为27.7°和47.2°处分别对应于ZnIn2S4(102)和(110)晶面. 对比O-ZIS/BP与O-ZIS样品的XRD谱图,可以看到二者的出峰位置和峰高没有明显的变化,表明BP的复合没有改变O-ZIS的晶体结构. 谱图中没有BP的特征峰出现,可能是由于BP含量少,未达到检测限.
2.2 O-ZIS/BP样品的微观形貌分析
采用扫描电镜(SEM)和透射电镜(TEM)观察样品的形貌结构. 从低倍SEM(图2a)图中可以看出,超声分散后的BP呈现纳米片状形貌;所制备的O-ZIS(图2b)纳米片是呈类花状结构,可以清楚地观察到它是由很多个纳米片聚合组装成的. 图2c为复合样品的SEM图,可以看到BP纳米片均匀地附着在O-ZIS纳米微球表面. 通过TEM(图2d)可以清楚地看到O-ZIS/BP复合样品超薄层状纳米片结构;从高分辨透射电镜(HRTEM)(图2e)中可以清晰地看到O-ZIS和BP纳米片之间紧密的界面接触,还可以清晰地观察到晶格条纹,晶格间距为0.33 nm对应于O-ZIS的(102)晶面,0.37 nm的晶格间距则对应于BP纳米片的(111)晶面. 这一结果也证实了O-ZIS纳米薄片与BP纳米片的成功复合.
2.3 O-ZIS/BP样品的XPS分析
用X射线光电子能谱(XPS)分析了O-ZIS/BP样品的表面组成、化学状态以及元素之间的相互作用. XPS全谱图(图3a)显示,样品含有Zn、In、O、S、P元素,表明BP与O-ZIS的成功复合. 从S 2p 的高分辨XPS谱图(图3b)可见,在结合能为161.5 eV和162.7 eV处的两个特征峰分别归属于S 2p3/2 和S 2p1/2,是S2-离子的特征峰[28];In 3d高分辨XPS谱图(图3c)显示位于444.5 eV和452.0 eV两个特征峰,分别对应于In 3d5/2、In 3d3/2的结合能;图3d为Zn 2p高分辨XPS谱图,特征峰位于1021.4 eV和1043.0 eV,分别属于Zn 2p3/2和Zn 2p1/2结合能,为正二价锌[29];样品的P 2p高分辨能谱(图3e)呈现出结合能133.2 eV的特征峰,通过分析得出特征峰归属于P-O键,可能是BP中的P与O-ZIS中的O成键[30]. XPS表征结果证明BP与O-ZIS的复合是化学键作用,而非简单的机械混合.
2.4 光催化降解四环素性能
通过在可见光(λ≥420 nm)照射下降解TC考察了样品的光催化性能. 在没有光照的条件下,先进行了30 min暗吸附,让体系达到吸附平衡. 如图4(a)所示,TC只是在光照而无催化剂条件下是不会发生自分解的,必须要在催化剂和光照共同作用下,才能实现TC的降解;相较于O-ZIS样品,O-ZIS/BP样品对TC的吸附能力略有增加,这可能是由于O-ZIS样品在复合BP后比表面积有所增加. 另外,在可见光照射下,复合材料降解TC的能力显著提高. 对比不同BP复合量的O-ZIS/BP催化剂(O-ZIS、O-ZIS/0.09% BP、O-ZIS/0.23% BP、O-ZIS/0.33% BP和O-ZIS/0.5% BP)的催化活性可以看出,BP的复合量为0.5%时,降解效果最佳,光照20 min,TC降解率达到90%,远高于空白O-ZIS样品的60%.
继续延长光照时间到40 min,TC降解率达到99%. 而继续增加BP负载量使得O-ZIS表面被覆盖,纳米片的活性位点减少,从而限制了其光催化性能. 这一结果表明BP的复合能极大提高ZIS光催化降解TC的活性.
由于TC的降解从实验方法上可以看出TC浓度仅仅是通过对特征峰值的大小来判断,但是在实际反应中,降解不是一个一步矿化的过程,反应过程中产生的自由基不断攻击四环素分子上的化学键,逐渐将其矿化成H2O和CO2,因此评价催化剂的降解能力还需要通过总有机碳的测试来判断[31]. 如图4(b)所示,降解反应30 min后整体矿化能力与其降解活性趋势一致,O-ZIS/BP催化剂具有比O-ZIS更优的矿化率,TOC去除率达到了64.5%,表现出较好的矿化能力,降低了环境安全风险.
2.5 光催化降解四环素反应机理
为了进一步研究O-ZIS/BP光催化降解TC的路径、中间产物以及活性物种作用的基团和键能,理解复合物光催化降解四环素的机理,本工作采用HPLC-MS分析了最终反应产物. 中间产物的降解路径图及荷质比m/z值如图5所示. 四环素具有双键、酚键和氨基基团,这些基团的电子密度较高,易受到·O2−和·OH自由基的攻击然后断开,尤其是双键. 在催反应的最初,由于N-C的键能较低,N—CH3之间的键会首先被断开,形成荷质比m/z = 431的化合物. 另一个反应路径主要是羟基化反应过程,首先是一个羟基基团攻击四环素分子,形成m/z = 461的化合物,然后羟基基团继续攻击,生成其他的分子化合物. 从图5中可以清楚的看出,在可见光照射下,四环素逐渐降解为一些分子量越来越小的中间体,如m/z为475、294、225、211的分子. 在整个降解过程中四环素的4个环被一步步打开,最终被矿化成CO2、H2O和无机离子[32].
为了进一步探究O-ZIS/BP在可见光照射下反应体系中产生的活性氧物种,采用电子自旋共振(EPR)对反应体系进行了表征. 实验以DMPO为自由基捕获剂,分别对于羟基自由基(·OH)和超氧自由基(·O2−)进行测试,结果如图6所示. 黑暗条件下在催化剂的甲醇分散液中未发现明显的EPR信号,如图6(a)所示,在黑暗条件下,催化剂的水分散体系中未检测到明显的DMPO-·OH特征信号,而在光照射条件下,出现明显的 DMPO-·OH特征峰信号,表明O-ZIS/BP在光催化降解过程中产生了·OH自由基. 此外,如图6(b)所示,在光照5 min后,出现的DMPO-·O2−特征峰信号,归属于·O2−活性物质的产生,表明溶液中O2被O-ZIS/BP的光生电子还原生成·O2−自由基,并作为主要活性物种参与光催化降解反应. 因此,EPR表征结果说明O-ZIS/BP光催化过程产生了·OH和·O2−自由基,在降解TC过程中起到了主要作用.
由于光催化反应过程中光会激发半导体价带上的电子,因此可以通过电化学的表征来研究光催化剂的电荷分离效率. 其中,光电流测试是评估光生载流子分离和传输效率的方法之一. 本实验通过光电流测试表征了样品光生电子的分离效率,结果如图7所示,空白O-ZIS样品的光电流响应强度较小,说明O-ZIS的电子-空穴分离效率比较低;O-ZIS/BP复合样品的光电流响应强度有明显增加,大约是O-ZIS空白样品的3倍,且经过7轮开关灯循环,其光电流强度仍能保持较高的强度,说明对O-ZIS进行BP复合可以显著提高其光生载流子的分离效率,有利于光催化性能的提高[33].
基于以上表征结果分析, O-ZIS/BP复合物提高光催化降解TC活性的可能机理,如图8所示. 前期报道过O-ZIS的导带(CB)和价带(VB)电势分别是-1.72 eV和0.76 eV[11],并利用DRS测得的样品吸收带边计算出O-ZIS的禁带宽度为2.48 eV;另外,文献报道剥离的BP纳米片的导带(CB)和价带(VB)电势分别是- 0.63 eV和1.75 eV,表明BP能很好地与O-ZIS实现能带匹配,形成异质结[34]. 因此,在可见光照射下,O-ZIS和BP中的价带电子均被激发至导带,BP导带电子与O-ZIS价带空穴通过致密的相界面复合,实现了O-ZIS和BP自身电子空穴对的有效分离,从而极大提高了其光催化活性. BP价带空穴氧化异质结表面吸附的水产生了·OH,O-ZIS导带电子还原O2产生·O2-,这些具有强氧化能力的自由基将TC氧化降解. 可以看出,O-ZIS/BP复合物光催化降解四环素(TC)反应过程如下:
O-ZIS+hν→e−+h+ (3) BP+hν→e−+h+ (4) e−+O2→⋅O−2自由基 (5) h++OH−→⋅OH自由基 (6) 有机污染物+⋅O−2自由基→降解产物 (7) 有机污染物+⋅OH自由基→降解产物 (8) h++e−→复合 (9) 3. 结论(Conclusion)
通过水热法合成O-ZIS纳米片,再通过静电吸附作用将超声剥离的BP纳米片成功复合在O-ZIS纳米片表面,形成O-ZIS/BP复合催化剂. BP的复合没有改变O-ZIS的晶体结构,BP纳米片能与O-ZIS纳米片形成紧密的界面接触,且形成了P—O化学键. 复合样品在可见光下表现出优异的光催化降解四环素性能,BP的复合量对O-ZIS材料的光催化性能也有影响,O-ZIS/0.5%wt BP样品的降解活性最佳,反应20 min,TC降解率达到90%,矿化率达到64.5%,远高于空白O-ZIS样品. 光电流测试结果表明BP纳米片的复合能显著促进光生电子和空穴对的分离效率. ·O2−和·OH自由基在O-ZIS/BP光催化降解反应过程中起到主要作用,能实现四环素的开环分解,直至完全矿化. 该结果将为开发通过BP纳米片复合光催化剂的方法来提高光催化活性提供新的思路.
-
-
[1] QIAO M, YING G G, SINGER A C, et al. Review of antibiotic resistance in China and its environment [J]. Environment International, 2018, 110: 160-172. doi: 10.1016/j.envint.2017.10.016 [2] 郝迪. 抗生素在水环境中的生态效应及危害防御 [J]. 现代农村科技, 2019(4): 101. doi: 10.3969/j.issn.1674-5329.2019.04.086 HAO D. Ecological effects and hazard prevention of antibiotics in aquatic environment [J]. Xiandai Nongcun Keji, 2019(4): 101(in Chinese). doi: 10.3969/j.issn.1674-5329.2019.04.086
[3] 赵孟欣, 孟哲, 李和平, 等. 氧化石墨烯调控钼酸铋在可见光下选择性光催化降解环境水中的抗生素 [J]. 高等学校化学学报, 2020, 41(11): 2479-2487. doi: 10.7503/cjcu20200423 ZHAO M X, MENG Z, LI H P, et al. Photodegradation of antibiotic in environmental water by graphene oxide modulation bismuth molybdate under visible light irradiation [J]. Chemical Journal of Chinese Universities, 2020, 41(11): 2479-2487(in Chinese). doi: 10.7503/cjcu20200423
[4] 穆容心, 李丽媛, 邵芸, 等. TiO2纳米棒对四环素的光催化降解 [J]. 环境化学, 2010, 29(3): 476-480. MU R X, LI L Y, SHAO Y, et al. Photocatalytic tetracycline degradation over tio2 nanorods [J]. Environmental Chemistry, 2010, 29(3): 476-480(in Chinese).
[5] LIU H, WANG C Y, Wang G X. Photocatalytic advanced oxidation processes for water treatment: Recent advances and perspective [J]. Chemistry, an Asian Journal, 2020, 15(20): 3239-3253. doi: 10.1002/asia.202000895 [6] CHENG M, ZENG G M, HUANG D L, et al. Hydroxyl radicals based advanced oxidation processes (AOPs) for remediation of soils contaminated with organic compounds: A review [J]. Chemical Engineering Journal, 2016, 284: 582-598. doi: 10.1016/j.cej.2015.09.001 [7] 王帅, 王振, 邱俊杰, 等. 零维、一维、二维无机纳米材料催化还原CO2研究进展 [J]. 功能材料, 2018, 49(12): 12071-12078. doi: 10.3969/j.issn.1001-9731.2018.12.010 WANG S, WANG Z, QIU J J, et al. Research progress of 0D, 1D and 2D inorganic nanomaterials as catalysts for the reduction of carbon dioxide [J]. Journal of Functional Materials, 2018, 49(12): 12071-12078(in Chinese). doi: 10.3969/j.issn.1001-9731.2018.12.010
[8] FU Q, BAO X H. Surface chemistry and catalysis confined under two-dimensional materials [J]. Chemical Society Reviews, 2017, 46(7): 1842-1874. doi: 10.1039/C6CS00424E [9] PAN B, WU Y, QIN J N, et al. Ultrathin Co0.85Se nanosheet cocatalyst for visible-light CO2 photoreduction [J]. Catalysis Today, 2019, 335: 208-213. doi: 10.1016/j.cattod.2018.11.017 [10] KHAN K, TAREEN A K, ASLAM M, et al. Recent developments in emerging two-dimensional materials and their applications [J]. Journal of Materials Chemistry C, 2020, 8(2): 387-440. doi: 10.1039/C9TC04187G [11] WANG S B, WANG Y, ZHANG S L, et al. Supporting ultrathin ZnIn2S4 nanosheets on Co/N-doped graphitic carbon nanocages for efficient photocatalytic H2 generation [J]. Advanced Materials, 2019, 31(41): 1903404. doi: 10.1002/adma.201903404 [12] PAN B, WU Y, RHIMI B, et al. Oxygen-doping of ZnIn2S4 nanosheets towards boosted photocatalytic CO2 reduction [J]. Journal of Energy Chemistry, 2021, 57: 1-9. doi: 10.1016/j.jechem.2020.08.024 [13] LIU T Y, WANG C X, WANG W, et al. The enhanced performance of Cr(VI) photoreduction and antibiotic removal on 2D/3D TiO2/ZnIn2S4 nanostructures [J]. Ceramics International, 2021, 47(12): 17015-17022. doi: 10.1016/j.ceramint.2021.03.007 [14] LEI Z, YOU W, LIU M, et al. Photocatalytic water reduction under visible light on a novel ZnIn2S4 catalyst synthesized by hydrothermal method [J]. Chemical Communications, 2003(17): 2142-2143. doi: 10.1039/b306813g [15] PHILBROOK A, BLAKE C J, DUNLOP N, et al. Demonstration of co-polymerization in melamine-urea-formaldehyde reactions using 15N NMR correlation spectroscopy [J]. Polymer, 2005, 46(7): 2153-2156. doi: 10.1016/j.polymer.2005.01.031 [16] BROWN E N, KESSLER M R, SOTTOS N R, et al. In situ poly(urea-formaldehyde) microencapsulation of dicyclopentadiene [J]. Journal of Microencapsulation, 2003, 20(6): 719-730. doi: 10.3109/02652040309178083 [17] HUANG T, CHEN W, LIU T Y, et al. ZnIn2S4 hybrid with MoS2: A non-noble metal photocatalyst with efficient photocatalytic activity for hydrogen evolution [J]. Powder Technology, 2017, 315: 157-162. doi: 10.1016/j.powtec.2017.03.054 [18] ZHU M S, ZHAI C Y, FUJITSUKA M, et al. Noble metal-free near-infrared-driven photocatalyst for hydrogen production based on 2D hybrid of black Phosphorus/WS2 [J]. Applied Catalysis B:Environmental, 2018, 221: 645-651. doi: 10.1016/j.apcatb.2017.09.063 [19] MORITA A. Semiconducting black phosphorus [J]. Applied Physics A, 1986, 39(4): 227-242. doi: 10.1007/BF00617267 [20] WARSCHAUER D. Electrical and optical properties of crystalline black phosphorus [J]. Journal of Applied Physics, 1963, 34(7): 1853-1860. doi: 10.1063/1.1729699 [21] BRIDGMAN P W. Two new modifications of phosphorus [J]. Journal of the American Chemical Society, 1914, 36: 1344-1363. doi: 10.1021/ja02184a002 [22] KEYES R W. The electrical properties of black phosphorus [J]. Physical Review, 1953, 92(3): 580-584. doi: 10.1103/PhysRev.92.580 [23] SUN Z B, XIE H H, TANG S Y, et al. Ultrasmall black phosphorus quantum dots: Synthesis and use as photothermal agents [J]. Angewandte Chemie (International Ed. in English), 2015, 54(39): 11526-11530. doi: 10.1002/anie.201506154 [24] CHEN P F, LI N, CHEN X Z, et al. The rising star of 2D black phosphorus beyond graphene: Synthesis, properties and electronic applications [J]. 2D Materials, 2017, 5(1): 014002. doi: 10.1088/2053-1583/aa8d37 [25] 郭桂全, 胡巧红, 王承林, 等. g-C3N4/RGO的制备、光催化降解性能及其降解机理 [J]. 环境化学, 2021, 40(3): 808-817. doi: 10.7524/j.issn.0254-6108.2019092605 GUO G Q, HU Q H, WANG C L, et al. Preparation, photocatalytic degradation performance and degradation mechanism of g-C3N4/RGO [J]. Environmental Chemistry, 2021, 40(3): 808-817(in Chinese). doi: 10.7524/j.issn.0254-6108.2019092605
[26] PAN B, LUO S J, SU W Y, et al. Photocatalytic CO2 reduction with H2O over LaPO4 nanorods deposited with Pt cocatalyst [J]. Applied Catalysis B:Environmental, 2015, 168/169: 458-464. doi: 10.1016/j.apcatb.2014.12.046 [27] QIN J N, ZHAO Q, ZHAO Y L, et al. Metal-Free Phosphorus-Doped ZnIn2S4 Nanosheets for Enhanced Photocatalytic CO2 Reduction [J]. The Journal of Physical Chemistry, 2021, 125(43): 23813-23820. [28] WANG S, GUAN B Y, LOU X W D, Construction of ZnIn2S4–In2O3 Hierarchical Tubular Heterostructures for Efficient CO2 Photoreduction[J]. Journal of the American Chemical Society, 2018, 140(15), 5037-5040. [29] ELBANNA O, ZHU M S, FUJITSUKA M, et al. Black Phosphorus Sensitized TiO2 mesocrystal photocatalyst for hydrogen evolution with visible and near-infrared light irradiation [J]. ACS Catalysis, 2019, 9(4): 3618-3626. doi: 10.1021/acscatal.8b05081 [30] YANG G, ZHU Y A, LIANG Y J, et al. Crystal defect-mediated{0 1 0}facets of Bi2MoO6 nanosheets for removal of TC: Enhanced mechanism and degradation pathway [J]. Applied Surface Science, 2021, 539: 148038. doi: 10.1016/j.apsusc.2020.148038 [31] 赵真艺, 沈鑫怡, 陈定宁, 等. NH2-nFe3O4@ZnO@Ce磁性复合材料的制备及其对三氯酚污染物的光催化降解 [J]. 环境化学, 2020, 39(3): 643-652. doi: 10.7524/j.issn.0254-6108.2019080501 ZHAO Z Y, SHEN X Y, CHEN D N, et al. Preparation of NH2-nFe3O4@ZnO@Ce magnetic composite material and its photo-catalytic degradation of 2, 4, 6-trichlorophenol [J]. Environmental Chemistry, 2020, 39(3): 643-652(in Chinese). doi: 10.7524/j.issn.0254-6108.2019080501
[32] 袁文辉, 夏自龙, 李莉. N掺杂ZnIn2S4光催化剂的制备及可见光催化性能 [J]. 功能材料, 2014, 45(12): 12117-12121,12127. doi: 10.3969/j.issn.1001-9731.2014.12.023 YUAN W H, XIA Z L, LI L. Preparation and photocatalytic performance of N-doped ZnIn2S4 photocatalyst under visible light illumination [J]. Journal of Functional Materials, 2014, 45(12): 12117-12121,12127(in Chinese). doi: 10.3969/j.issn.1001-9731.2014.12.023
[33] 杨利伟, 刘丽君, 夏训峰, 等. pg-C3N4/BiOBr/Ag复合材料的制备及其光催化降解磺胺甲噁唑 [J]. 环境科学, 2021, 42(6): 2896-2907. YANG L W, LIU L J, XIA X F, et al. Preparation of pg-C3N4/BiOBr/Ag composite and photocatalytic degradation of sulfamethoxazole [J]. Environmental Science, 2021, 42(6): 2896-2907(in Chinese).
[34] RAN J R, ZHU B C, QIAO S Z. Phosphorene co-catalyst advancing highly efficient visible-Light photocatalytic hydrogen production [J]. Angewandte Chemie , 2017, 56(35): 10373-10377. doi: 10.1002/anie.201703827 期刊类型引用(1)
1. 马同宇,张雪,郭盛祺. WO_3/BiOBr复合材料对磷酸氯喹的光催化降解. 环境化学. 2024(11): 3863-3872 .  本站查看
本站查看
其他类型引用(1)
-






 下载:
下载: