-
近年来,由于农业灌溉、化肥使用、工业“三废”和生活污水排放,地下水和地表水中的NO3−-N污染令人担忧。在我国山东潍坊,饮用水井中NO3−-N质量浓度高达150 mg·L−1 [1],石家庄市浅层地下水NO3−-N质量浓度更是高达184 mg·L−1[2],同时我国部分河流也出现NO3−-N污染问题[3]。NO3−-N污染不仅会引起水体富营养化、生物多样性丧失等生态环境问题[4],还会对人类健康产生潜在威胁,如引发癌症和高铁血红蛋白血症等[5]。
目前,去除NO3−-N的方法主要有生物反硝化法、离子交换法、膜法、催化还原法等。其中,催化还原法具有转化率高、反应速率快等特点,是一种很有前途的NO3−-N脱除技术[6]。在众多催化还原材料中,纳米零价铁由于比表面积大、反应活性高、毒性低等优点已被运用到NO3−-N去除领域,但其尺寸小、表面能高、加上固有的磁性,使其易于团聚,导致表面活性位减少,进而降低NO3−-N的还原效率[7]。为了解决上述问题,国内外学者提出了一些改进方法。例如,以沸石、凹凸棒土、生物炭、聚合树脂等为载体制备负载型纳米零价铁,抑制纳米零价铁的团聚;在纳米零价铁中引入Pd、Pt、Ag、Au等贵金属和其他过渡金属(Al、Ni、Cu等),提高反应速率、减少氨氮生成量等[8-10]。
本研究以果壳活性炭为基体,采用硼氢化钠还原法一步制备铁/镍双金属负载活性炭(AC-Fe/Ni),优化了活性炭与金属铁、镍的最佳掺杂比,考察了初始pH、AC-Fe/Ni投加量、NO3−-N初始质量浓度、共存离子和有机物对NO3−-N去除效果和反应产物的影响;分析了AC-Fe/Ni去除NO3−-N过程中含氮物种的浓度变换,探究了AC-Fe/Ni还原NO3−-N的机理。同时,采用次氯酸钠氧化法去除出水中的氨氮,达到有效控制NO3−-N污染的目的。
-
主要试剂:七水硫酸亚铁(FeSO4·7H2O)、六水氯化镍(NiCl2·6H2O)、硼氢化钠(NaBH4)、无水乙醇、硝酸钾(KNO3)、亚硝酸钠(NaNO2)、氯化铵(NH4Cl)、乙酸、氯化钠(NaCl)、氯化钾(KCl)、氯化镁(MgCl2)、碳酸氢钠(NaHCO3)、磷酸二氢钾(KH2PO4)、硫酸钠(Na2SO4)、氨基磺酸、盐酸、氢氧化钠、氨基苯磺酰胺、N-1-萘乙二胺盐酸盐、碘化钾(KI)、碘化汞(HgI2)、酒石酸钾钠、5%次氯酸钠溶液。以上化学试剂均为分析纯,实验用水为去离子水。
主要仪器:火焰原子吸收分光光度计、气浴恒温振荡箱、电子天平、离心机、鼓风干燥箱、真空干燥箱、紫外可见分光光度计、真空封口机、破碎机。
-
将购买的果壳活性炭(AC)清洗至中性,烘干、破碎,过80目筛网,取筛下物备用,记作AC。按照一定比例称取FeSO4·7H2O、NiCl2·6H2O和AC于50 mL锥形瓶中,加入15 mL脱氧水溶解,超声10 min后于气浴恒温振荡箱中振荡20 min。随后,称取过量NaBH4溶于水中,缓慢滴入前驱液并继续反应30 min。之后,用脱氧水、无水乙醇洗涤至中性,在60 ℃真空干燥箱中干燥6 h,即得铁/镍双金属负载活性炭(AC-Fe/Ni)。载铁活性炭(AC-Fe)的制备方法同上。
-
用去离子水配置KNO3溶液作为模拟废水。批量实验在250 mL锥形瓶中进行,具体操作如下:在锥形瓶中加入100 mL已知浓度的NO3−-N溶液(20、30、40、50 mg·L−1),随后加入适量材料(1、2、3 g·L−1),放入180 r·min−1、25 ℃气浴恒温振荡箱中反应60 min,移取少量溶液过0.45 µm滤膜,测定反应后NO3−-N、NO2−-N和NH4+-N浓度。溶液初始pH(4、6、8、10)用1 mol·L−1盐酸和1 mol·L−1氢氧化钠调节,并在最优条件下考察共存阴离子(Cl−、SO42−、HCO3−和PO42−)、共存阳离子(Na+、K+、Mg2+)和有机物对NO3−-N去除效果的影响。
在AC-Fe/Ni还原NO3−-N的最优条件下,将处理出水过0.45 um滤膜。投加不同体积质量浓度的次氯酸钠,调节pH至7,反应一段时间后测量溶液中剩余NH4+-N浓度。
-
利用紫外可见分光光度计,按照国标HJ/T346-2007方法测定NO3−-N的浓度;按照HJ/353-2009方法测定NH4+-N浓度;按照GB7493-87测定NO2−-N的浓度。采用火焰原子吸收分光光度计法测定溶液中金属离子浓度。
根据上述方法得到的实验数据,利用式(1)计算NO3−-N的去除率R,利用式(2)、式(3)和式(4)分别计算反应后N2、NH4+-N和NO2−-N的选择性。根据式(5)和式(6)进行一级动力学和二级动力学拟合。
式中:R为NO3−-N的去除率,%;S为反应后的氮物种选择性,%;
$ {C}_{0} $ 为NO3−-N的初始质量浓度,mg·L−1;$ {C}_{t} $ 为t时刻NO3−-N质量浓度,mg·L−1;$ {C}_{\left[{\mathrm{N}\mathrm{H}}_{4}\right]} $ 为NH4+-N质量浓度,mg·L−1;$ {C}_{\left[{\mathrm{N}\mathrm{O}}_{2}\right]} $ 为NO2−-N质量浓度,mg·L−1。式中:t为反应时间,min;k1为一级动力学拟合反应速率常数,min−1;k2为二级动力学拟合反应速率常数,g·(L·min)−1。
-
利用日本电子JSM-7001F 扫描电镜(SEM)观察AC和AC-Fe/Ni的表面形貌。利用日本理学Ultima IV型X-射线衍射仪(XRD)分析样品的晶体结构,辐射源为Cu-Kα,衍射角为5°~85°。利用美国热电ESCALAB 250Xi型X射线光电子能谱 (XPS)分析样品表面元素组成和价态,辐射源为Al-Kα,以C1s 的结合能为284.8 eV标准进行荷电校正。
-
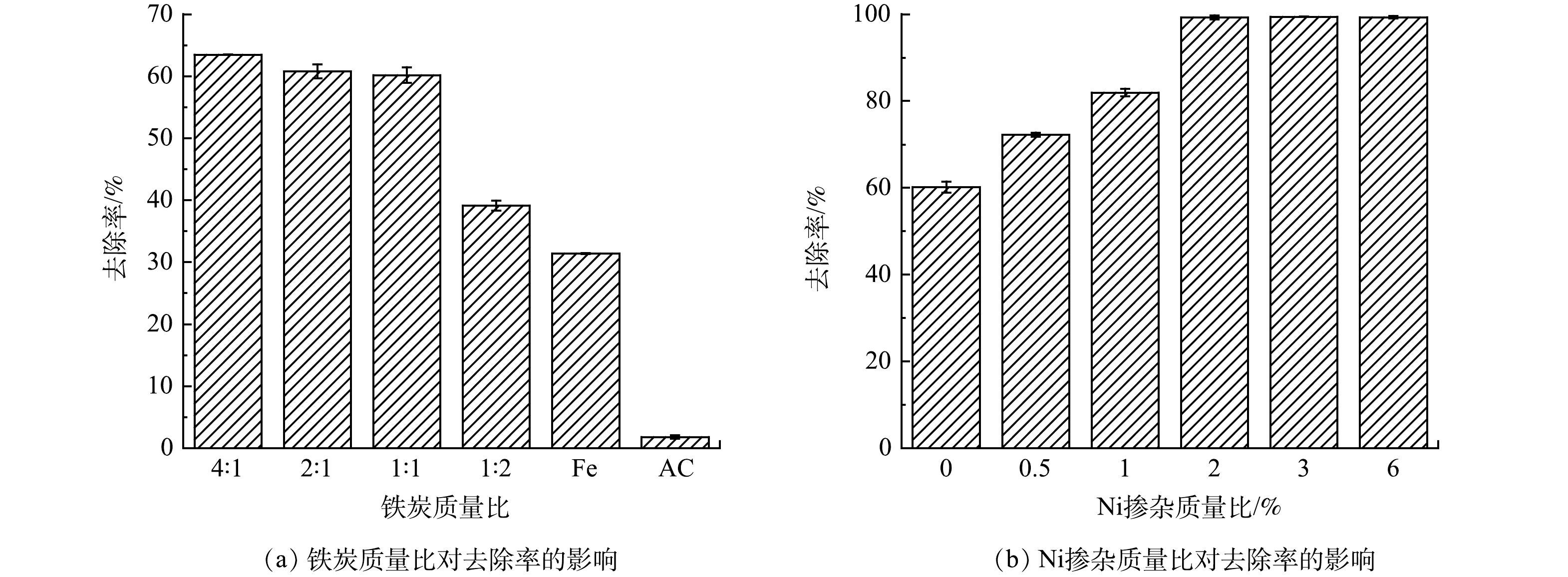
1)铁炭质量比对NO3−-N去除率的影响。铁炭质量比对NO3−-N去除效果的影响结果如图1(a)。可以看出,仅投加Fe和AC时,NO3−-N去除率分别为31.42%和1.80%。相比较,将Fe负载到AC上,NO3−-N的去除效果得到明显提升,这归因于AC对Fe的分散作用,使AC-Fe能够暴露更多的活性位点[11]。当铁炭质量比由1:2提升到1∶1时,NO3−-N的去除率由39.14%迅速增加到60.18%。之后继续提高Fe的含量,NO3−-N的去除率提升较小,因此后续实验固定铁炭质量为1∶1。
2) Ni掺杂对NO3−-N去除率的影响。为探究Ni掺杂对NO3−-N去除效果的影响,制备了Ni掺杂质量比分别为0%、0.5%、1%、2%、3%和6%的AC-Fe/Ni材料。由图1(b)可知,Ni的引入明显提高了NO3−-N的去除率。随着Ni的掺杂比由0%上升到2%,NO3−-N的去除率由60.18%不断增加到99.29%,较AC-Fe提高约40%。之后进一步提高Ni的含量,NO3−-N的去除率几乎不变,因此选择2%作为最佳Ni掺杂比。
-
1)扫描电镜 (SEM) 分析。图2为AC和AC-Fe/Ni的SEM图。可以看到,AC呈大小不规则的块状,表面较光滑。在负载纳米零价Fe和Ni后,块状AC 被大量蓬松的球形颗粒物包裹,其直径约50~80 nm,表明纳米Fe和Ni已成功负载。
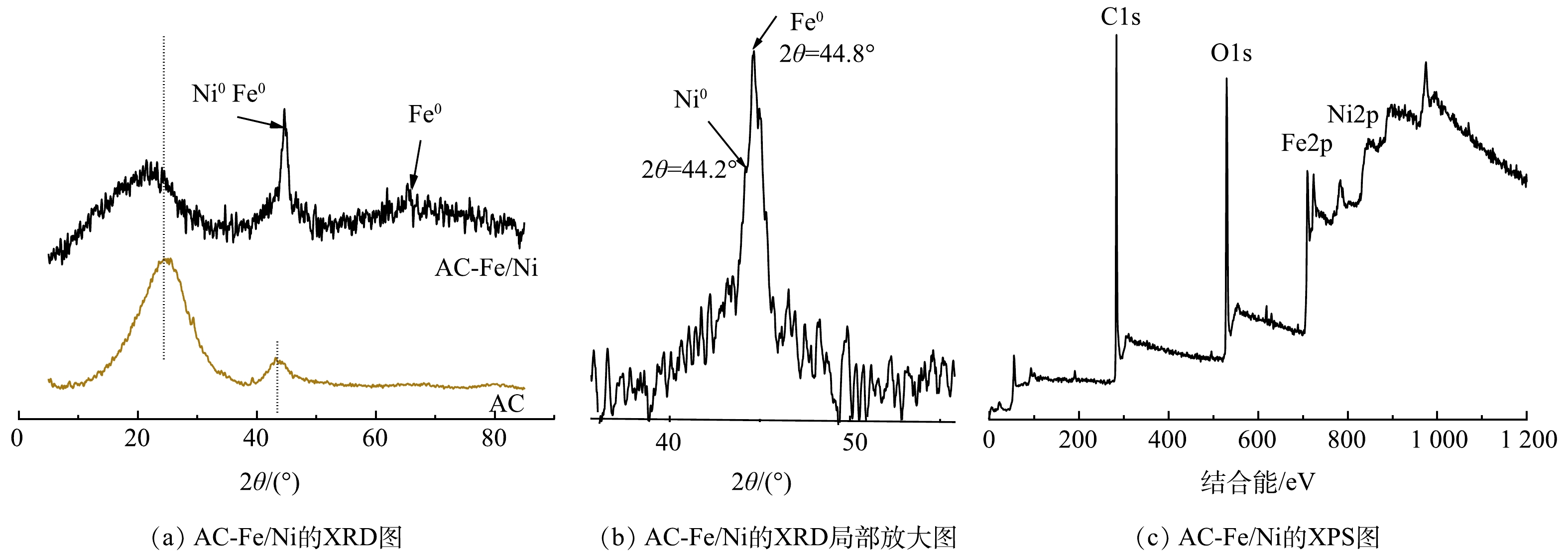
2) XRD与XPS分析。图3(a)为AC和AC-Fe/Ni的XRD图。在AC的XRD图中,出现在24.7°和43.3°的衍射峰为活性炭的碳微晶结构特征峰。相比较,在AC-Fe/Ni在44.8 °和65.3 °处出现了新的衍射峰,分别属于α-Fe0 的(110)晶面和(200) 晶面,说明Fe0的成功负载[12]。Ni0的特征衍射峰位于44.2°,因其含量较低且峰位置与α-Fe0的峰位置叠加不能明显区分[13]。在AC-Fe/Ni的XRD图中,未见铁氧化物和铁镍氧化物的特征峰,说明Fe、Ni均以单质形式存在。与此同时,碳微晶结构的特征峰强度显著降低,归因于纳米Fe0和Ni0对AC的覆盖。图3(c)为AC-Fe/Ni的XPS图。可以看出,在284.6、530.1、724.1和856.6 eV处分别出现了C1s、O1s、Fe2p和Ni2p的特征峰,进一步确认铁与镍成功负载在AC表面。
-
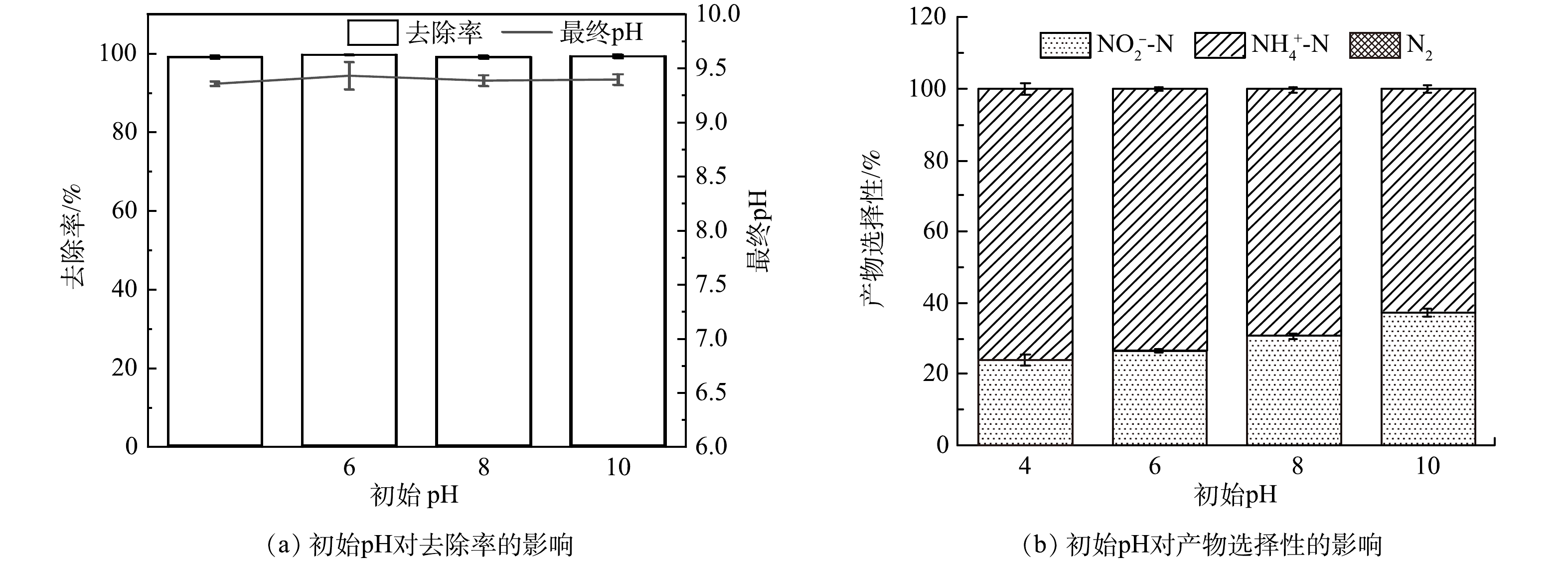
1)不同初始pH的影响。不同初始pH对AC-Fe/Ni去除NO3−-N的影响如图4所示。可以看到,初始pH的变化对NO3−-N的去除无明显影响,NO3−-N的去除率均保持在99%以上,说明AC-Fe/Ni能在较宽pH范围内有效去除NO3−-N。同时,溶液反应后的最终pH维持在9.3~9.4,表明初始pH的改变不会对最终pH产生明显影响。最终pH升高主要归因于去除NO3−-N的过程消耗了H+,同时Fe0的腐蚀会产生OH− [14]。从产物NO2−-N、NH4+-N和N2的选择性来看,NH4+-N和N2是主要的反应产物,而且随着初始pH由4升高到10,N2的选择性逐渐提高。这是因为在酸性条件下,溶液中丰富的H+增加了N-H之间碰撞的概率,使NO3−-N更容易向NH4+-N转换[15]。
鉴于溶液pH对金属离子的释出有重要影响,实验对反应后溶液中的总铁和总镍浓度进行了测定。结果显示,总铁质量浓度为0.18 mg·L−1,达到地下水二类水质量标准,总镍质量浓度低于测定方法检测下限0.05 mg·L−1,表明AC-Fe/Ni投入使用不会释放金属离子引起二次污染。
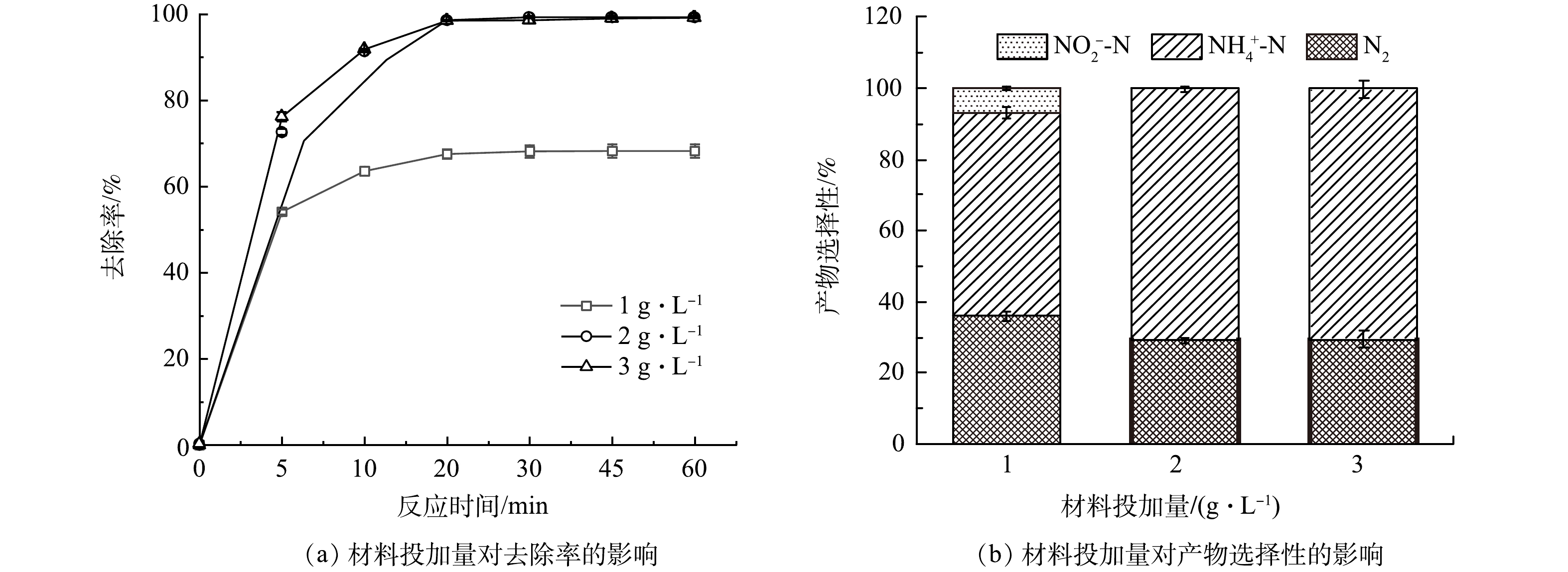
2) AC-Fe/Ni投加量的影响。AC-Fe/Ni投加量NO3−-N去除效果的影响如图5。当材料投加量为1 g·L−1时,NO3−-N的去除率仅有68.26%,这是因为材料投加量过少时活性位点不够,NO3−-N不能完全去除。当材料投加量增加到2 g·L−1时,NO3−-N的去除率达到99.30%;进一步增加投加量到3 g·L−1,NO3−-N去除率几乎不变。从反应后产物的选择性来看,当材料投加量1 g·L−1时,NO2−-N选择性达到6.93%,这是由于活性物种不足,导致反应后NO2−-N累积。不过,此时N2选择性达到35.96%,略高于AC-Fe/Ni投加量2 g·L−1和3 g·L−1时的结果。与此同时,AC-Fe/Ni投加量较少时会降低体系中H的生成速度和NO3−-N的加氢速度,减少N中间体与H结合,使得产物更易向N2转换[15]。
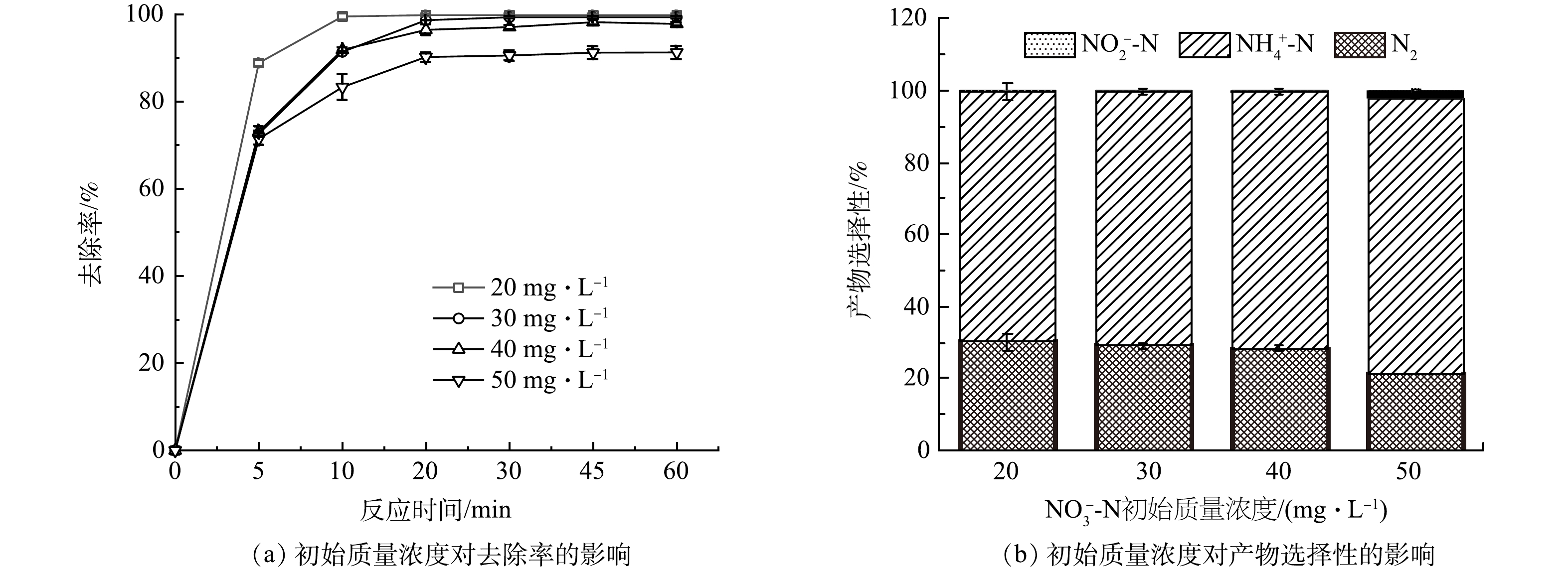
3) NO3−-N初始浓度的影响。NO3−-N初始质量浓度对AC-Fe/Ni去除NO3−-N的影响如图6所示。当NO3−-N质量浓度为20 mg·L−1和30 mg·L−1时,NO3−-N的去除率分别达到99.77%和99.29%;当NO3−-N质量浓度为40 mg·L−1时,NO3−-N的去除率为97.75%;当NO3−-N质量浓度为50 mg·L−1时,去除率降到91.21%。产物选择性分析结果表明,当NO3−-N质量浓度在20~40 mg·L−1时,反应后产物选择性无明显差别。当NO3−-N质量浓度为50 mg·L−1,由于NO3−-N浓度过高,反应活性位点不足,导致NO2−-N少量累积,N2选择性下降到21.15%。这与其他研究[16]结果一致,即当NO3−-N初始浓度过高时,会导致其降解效果以及N2选择性的略微下降。
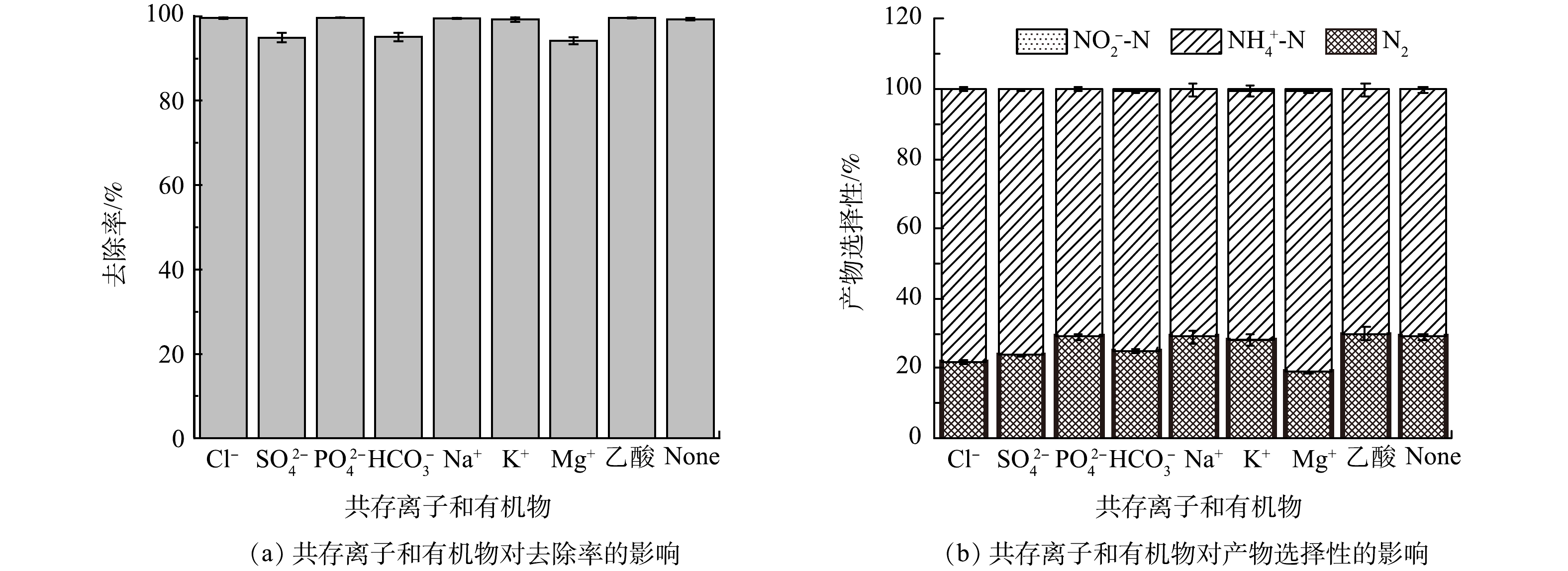
4)共存离子和有机物的影响。真实水环境中通常存在多种阴离子、阳离子和有机物,他们可能影响AC-Fe/Ni去除NO3−-N的效率。为此,本研究根据《地下水质量标准》(GBT 14848-2017) 和《地表水环境质量标准》(GB 3838-2002) 水标准,分别固定Cl−、SO42−和HCO3−的质量浓度为350 mg·L−1、总磷质量浓度为0.4 mg·L−1,Na+、K+、Mg2+和乙酸质量浓度为200 mg·L−1,在此条件下研究AC-Fe/Ni对NO3−-N的去除效果,结果如图7所示。在阴离子共存时,SO42−和HCO3−会导致AC-Fe/Ni对NO3−-N的去除率分别下降4.38%、4.22%。这是因为SO42−和HCO3−会在材料表面与Fe0形成FeSO4和FeCO3,导致材料表面钝化,阻碍了NO3−-N与金属纳米颗粒的接触和电子转移,从而降低反应活性[17-18]。从产物选择性来看,SO42−、HCO3− 和Cl−共存均会导致产物中N2选择性降低,但仍维持在20%以上。在本实验中,PO42−浓度较低,对NO3−-N的去除没有产生明显影响。在阳离子和有机物共存时,Na+、K+和乙酸对NO3−-N的去除率和N2选择性均无影响,仅Mg2+会使NO3−-N的去除率下降4.68%且会降低N2选择性。综上所述,阴离子、阳离子和有机物共存对AC-Fe/Ni去除NO3−-N的效果影响较小,AC-Fe/Ni具有较强的抗干扰能力。
5)反应动力学分析。采用一级和二级动力学模型对AC-Fe/Ni去除NO3−-N的动力学数据进行拟合,拟合参数见表1。二级动力学模型拟合得到的R2更大,表明AC-Fe/Ni去除NO3−-N的过程更符合二级动力学模型。同时,反应速率常数k2随着NO3−-N初始质量浓度的提高而降低,说明NO3−-N浓度的提高会降低反应速率,在低浓度下反应进程更快。
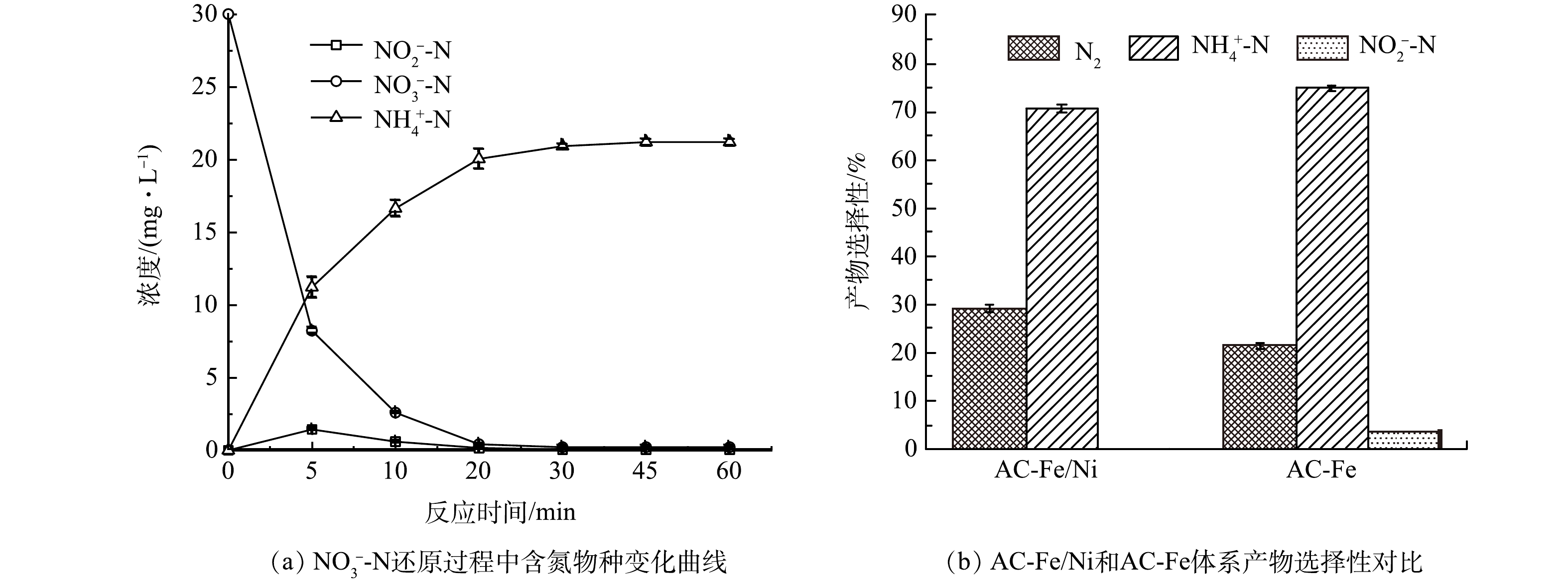
6)反应机理分析。为研究AC-Fe/Ni去除NO3−-N过程中含氮物种的转化过程,对不同反应时间体系中的NO3−-N、NO2−-N和NH4+-N浓度进行了测定,结果如图8(a)所示。可以看到,NO3−-N质量浓度在前20 min内从30 mg·L−1快速降至0.39 mg·L−1;NO3−-N质量浓度则先升高至1.43 mg·L−1,之后再降至0.05 mg·L−1;NH4+-N质量浓度随反应时间延长先逐渐升高,之后维持在21.17 mg·L−1左右。进一步的分析结果表明,反应5 min时总氮质量浓度已低于初始总氮质量浓度,说明在反应过程中有一部分氮被转换为N2挥发到空气中。与此同时,对比了AC-Fe/Ni与AC-Fe去除NO3−-N时产物的选择性,结果如图8(b)所示。可以看到,AC-Fe/Ni体系中N2的选择性提高了约8%,且反应后几乎无NO2−-N累积。基于以上结果, AC-Fe/Ni去除NO3−-N的机理可表述如下:Fe0作为主要的电子供体参与反应,将NO3−-N还原为NO2−-N、NH4+-N和N2。同时由于Fe0和Ni0之间存在氧化还原电位差,使得Fe0作为阳极Ni0作为阴极形成原电池加速电子从Fe0转向Ni0,提高了NO3−-N的去除效率。此外,这一过程也促进了Fe0产生的H2在Ni0表面形成原子氢 (Hads),Hads是一种强还原剂[19]。同时,Ni—Hads对NO2−-N具有较强的吸附性能和较高的反应活性,在Ni0表面能够促进NO2−-N还原为NH4+-N和N2[20]。不过,由于Ni0的含量较低,形成的Ni—Hads较少,且产生NH4+-N所需的Ni—Hads是产生N2所需量的3倍,因此,Ni—Hads更有利于N2的形成[21]。相关反应式参见式(7)~式(14)。
-
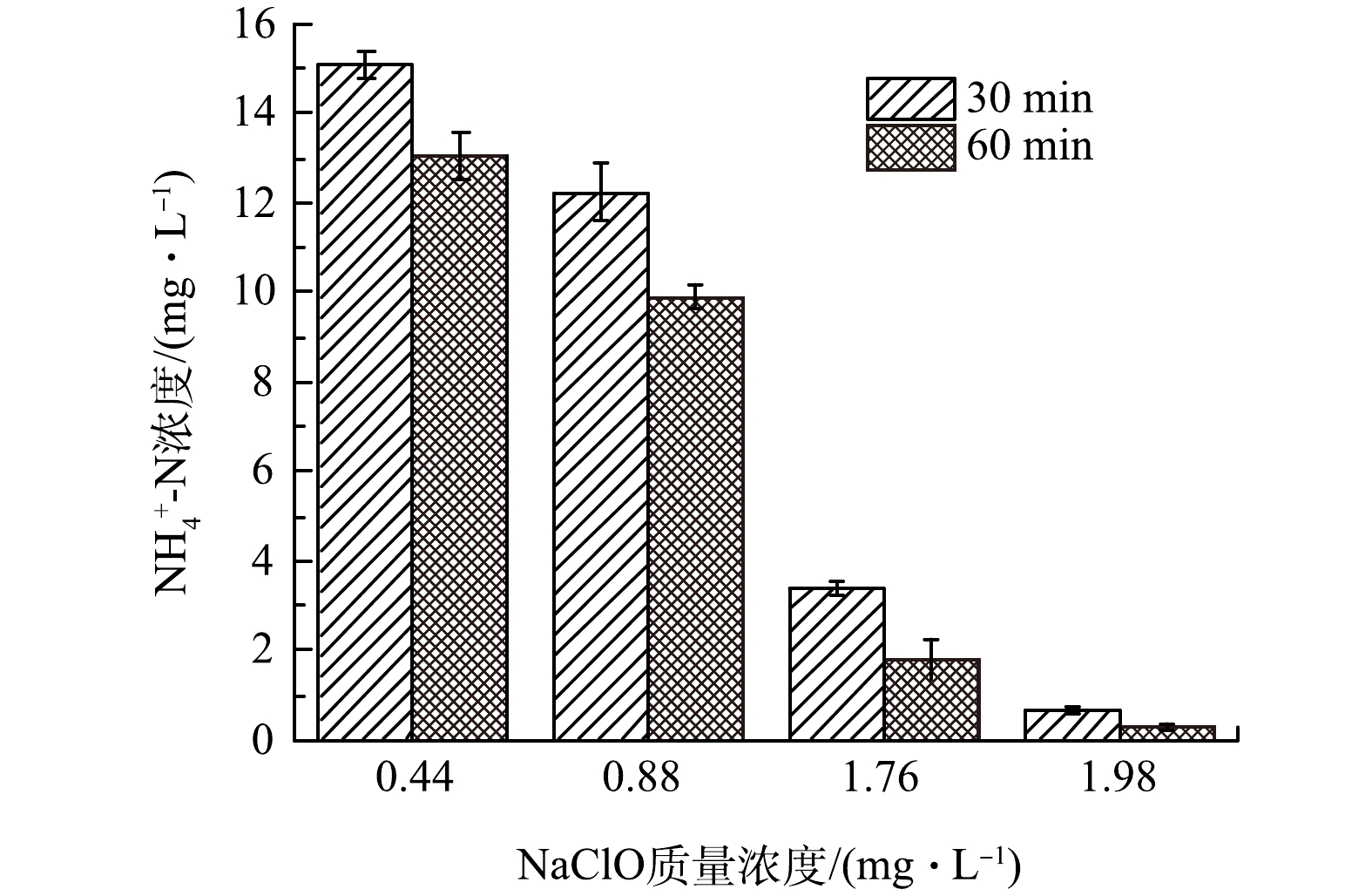
NaClO是常用的漂白剂和消毒剂。因其具有较强的氧化能力,可以将NH4+-N氧化成N2,已被用于水环境中NH4+-N的去除[22-23]。为此,本研究采用NaClO氧化法去除AC-Fe/Ni还原NO3−-N出水中的NH4+-N,结果如图9所示。随着NaClO投加量增加,NH4+-N浓度逐渐降低。当所投加的NaClO质量浓度为1.76 mg·L−1时,反应30 min后溶液中的NH4+-N质量浓度接近0.5 mg·L−1,反应60 min后降至0.28 mg·L−1,达到《地下水质量标准》 (GB/T 14848-2017) 三类标准限值。
-
1)该研究制备的AC-Fe/Ni能够快速、有效去除NO3−-N。AC-Fe/Ni的最佳制备条件为铁炭质量比1:1,镍掺杂质量比2%。Ni的掺杂不仅能够提高NO3−-N的去除效果,还能在一定程度上提高N2的选择性。在最优条件下,30 mg·L−1的NO3−-N在反应20 min后去除率为99.29 %,N2选择性为29.26%。
2)当NO3−-N初始质量浓度为30 mg·L−1,AC-Fe/Ni投加量为2 g·L−1时,AC-Fe/Ni能在较宽pH(4~10)范围内有效还原NO3−-N,碱性条件更有利于N2的形成。随着AC-Fe/Ni投加量降低,NO3−-N的去除率下降,但产物中N2的选择性有所提高;当NO3−-N初始质量浓度升至50 mg·L−1,NO3−-N去除率和产物中N2的选择性均有所降低,同时导致少量NO2−-N累积。
3) AC-Fe/Ni去除NO3−-N的过程符合二级反应动力学,反应速率常数k随NO3−-N初始质量浓度的提高而降低,产物为NH4+-N和N2。AC-Fe/Ni具有较好的抗干扰能力,共存阴离子、阳离子和有机物对NO3−-N的去除率和N2选择性影响较小。
4)次氯酸钠联合能有效去除NO3−-N还原出水中的NH4+-N,最终溶液中剩余NH4+-N仅为0.28 mg·L−1,达到《地下水质量标准》 (GB/T 14848-2017) 三类标准限值。
AC-Fe/Ni联合次氯酸钠去除水中硝酸盐氮
Removal of nitrate nitrogen from water by AC-Fe/Ni combining sodium hypochlorite
-
摘要: 随着工业、农业的快速发展,硝酸盐氮(NO3−-N)的污染范围不断扩大、污染程度不断加深,因此,有效去除水中的NO3−-N具有重要意义。本研究通过在活性炭上原位负载零价铁和镍,制备了载铁/镍双金属活性炭(AC-Fe/Ni),并联合次氯酸钠去除水中的NO3−-N。结果表明,AC-Fe/Ni能够快速还原NO3−-N,反应20 min时NO3−-N的去除率为99.29%,比AC-Fe提高40%,且N2选择性提高8%。AC-Fe/Ni在酸性和碱性条件下均能有效还原NO3−-N,但碱性条件更有利于形成N2。减少材料投加量或提高NO3−-N初始质量浓度均会降低NO3−-N的去除率;共存阴离子、阳离子、有机物对NO3−-N的去除无明显影响。NO3−-N的还原过程总铁释出量仅为0.28 mg·L−1,总镍浓度低于0.05 mg·L−1。与此同时,次氯酸钠联用能有效去除出水中的NH4+-N,反应60 min时溶液中残余氨氮仅为0.28 mg·L−1。Abstract: With the rapid development of industry and agriculture, the scope of nitrate nitrogen (NO3−-N) pollution continues to expand and the pollution degree becomes increasingly severe. Therefore, it is of great significance to effectively remove NO3−-N in water. In this study, iron/nickel bimetallic nanoparticles loaded activated carbon (AC-Fe/Ni) was prepared by a in-situ reduction method. Subsequently, the AC-Fe/Ni was combined with sodium hypochlorite for the NO3−-N removal. The results show that AC-Fe/Ni could quickly reduce NO3−-N. The removal rate of NO3−-N reached 99.29% after 20 min of reaction, which was 40% higher than the AC-Fe, and the N2 selectivity increased by 8%. AC-Fe/Ni could effectively reduce NO3−-N under both acidic and alkaline conditions, but alkaline condition was more conducive to the formation of N2. Reducing the AC-Fe/Ni dosage or increasing the initial NO3−-N concentration decreased the removal rate of NO3−-N. Coexist anions, cations and organic matter had no significant effect on the removal of NO3−-N. The total iron released in the solution was only 0.28 mg·L−1 during the reduction process of NO3−-N, and the total nickel concentration was less than 0.05 mg·L−1. At the same time, the combination of sodium hypochlorite could effectively remove NH4+-N in the effluent, and the residual NH4-N in the solution after 60 min of reaction was only 0.28 mg·L−1.
-
Key words:
- nitrate nitrogen /
- activated carbon /
- iron/nickel bimetal /
- sodium hypochlorite
-

-
表 1 AC-Fe/Ni去除NO3−-N的动力学拟合参数
Table 1. Kinetic fitting parameters of NO3−-N removal by AC-Fe/Ni
NO3−-N初始浓度/
(mg·L−1)一级动力学 二级动力学 k1/ min−1 R2 k2/(g·(L·min)−1) R2 20 0.305 0.865 1.173 0.950 30 0.184 0.968 0.070 0.927 40 0.163 0.920 0.035 0.976 50 0.108 0.862 0.009 0.998 -
[1] 徐春英, 李玉中, 李巧珍, 等. 山东潍坊地下水硝酸盐污染现状及δ^(15)N溯源[J]. 生态学报, 2011, 31(21): 6579-6587. [2] 赵俊玲, 曹李靖, 赵雪莲, 等. 石家庄市浅层地下水中硝酸盐氮污染现状及原因分析[J]. 洛阳工业高等专科学校学报, 2005, 15(2): 6-7,44. [3] ZHANG X, ZHANG Y, SHI P, et al. The deep challenge of nitrate pollution in river water of China[J]. Science Of The Total Environment, 2021, 770: 144674. doi: 10.1016/j.scitotenv.2020.144674 [4] BIJAY S, GRASWELL E. Fertilizers and nitrate pollution of surface and ground water: an increasingly pervasive global problem[J]. SN Applied Sciences, 2021, 3(4): 1-24. [5] SHI J L, YI S N, HE H L, et al. Preparation of nanoscale zero-valent iron supported on chelating resin with nitrogen donor atoms for simultaneous reduction of Pb and NO3−[J]. Chemical Engineering Journal, 2013, 230: 166-171. doi: 10.1016/j.cej.2013.06.088 [6] ABASCAL E, GOMEZ-COMA L, ORTIZ I, et al. Global diagnosis of nitrate pollution in groundwater and review of removal technologies[J]. Science of the Total Environment, 2022, 810: 152233. doi: 10.1016/j.scitotenv.2021.152233 [7] RYU A, JEONG S, JANG A, et al. Reduction of highly concentrated nitrate using nanoscale zero-valent iron: Effects of aggregation and catalyst on reactivity[J]. Applied Catalysis B- Environmental, 2011, 105(1-2): 128-135. doi: 10.1016/j.apcatb.2011.04.002 [8] 吕晓书, 王霞玲, 蒋光明, 等. 纳米零价铁基材料去除水中硝酸盐污染的研究进展[J]. 材料导报, 2023, 37(4): 58-67. [9] XU J, PU Y, QI W K, et al. Chemical removal of nitrate from water by aluminum-iron alloys[J]. Chemosphere, 2017, 166: 197-202. doi: 10.1016/j.chemosphere.2016.09.102 [10] LIOU Y H, LO S L, LIN C J, et al. Chemical reduction of an unbuffered nitrate solution using catalyzed and uncatalyzed nanoscale iron particles[J]. Journal of Hazardous Materials, 2005, 127(1-3): 102-110. doi: 10.1016/j.jhazmat.2005.06.029 [11] 汪虹西, 廖兵, 卢涛, 等. 零价铁-生物炭复合材料对地下水中硝酸盐的去除[J]. 环境工程学报, 2020, 14(12): 3317-3328. doi: 10.12030/j.cjee.201912045 [12] SHI L N, DU J H, CHEN Z L, et al. Functional kaolinite supported Fe/Ni nanoparticles for simultaneous catalytic remediation of mixed contaminants (lead and nitrate) from wastewater[J]. Journal of Colloid and Interface Science, 2014, 428: 302-307. doi: 10.1016/j.jcis.2014.04.059 [13] HE Y H, LIN H, DONG Y B, et al. Zeolite supported Fe/Ni bimetallic nanoparticles for simultaneous removal of nitrate and phosphate: Synergistic effect and mechanism[J]. Chemical Engineering Journal, 2018, 347: 669-681. doi: 10.1016/j.cej.2018.04.088 [14] KIM D G, HWANG Y H, SHIN H S, et al. Kinetics of nitrate adsorption and reduction by nano-scale zero valent iron (NZVI): Effect of ionic strength and initial pH[J]. KSCE Journal of Civil Engineering, 2016, 20(1): 175-187. doi: 10.1007/s12205-015-0464-3 [15] LV X S, PENG H H , WANG X L, et al. Nitrate reduction by nanoscale zero valent iron (nFe0)-based systems: Mechanism, reaction pathway and strategy for enhanced N2 formation[J]. Chemical Engineering Journal, 2022, 430: 4. [16] 邱思远, 高芳, 王金霞, 等. 生物炭零价铁去除水中硝酸盐氮影响因素探究[J]. 当代化工研究, 2023(11): 33-35. [17] LIU Y, PHENRAT T, LOWRY, G V. Effect of TCE concentration and dissolved groundwater solutes on NZVI-promoted TCE dechlorination and H2 evolution[J]. Environmental Science & Technology, 2007, 41(22): 7881-7887. [18] SHEN Z H, LIU D R, DONG X Y, et al. Nitrate reduction using iron and copper bimetallic nanoparticles supported by chelating resin: effect of solution chemistry, mechanism, and regeneration[J]. Journal of Environmental Engineering, 2020, 146(4): 04020011. doi: 10.1061/(ASCE)EE.1943-7870.0001671 [19] HOU M T, TANG Y, XU J, et al. Nitrate reduction in water by aluminum-iron alloy particles catalyzed by copper[J]. Journal of Environmental Chemical Engineering, 2015, 3(4): 2401-2407. doi: 10.1016/j.jece.2015.08.014 [20] LIU Y L, GONG X B, YANG W J, et al. Selective reduction of nitrate into nitrogen using Cu/Fe bimetal combined with sodium sulfite[J]. Industrial & Engineering Chemistry Research, 2019, 58(13): 5175-5185. [21] TANG T T, XING Q J, ZHANG S H, et al. High selective reduction of nitrate into nitrogen by novel Fe-Cu/D407 composite with excellent stability and activity[J]. Environmental Pollution, 2019, 252: 888-896. doi: 10.1016/j.envpol.2019.05.071 [22] 张胜利, 刘丹, 曹臣. 次氯酸钠氧化脱除废水中氨氮的研究[J]. 工业用水与废水, 2009, 40(3): 23-26. doi: 10.3969/j.issn.1009-2455.2009.03.006 [23] 方小琴, 胡君杰, 夏俊方. NaClO氧化去除高盐废水中氨氮的影响因素及其动力学研究[J]. 工业用水与废水, 2017, 48(2): 18-23. doi: 10.3969/j.issn.1009-2455.2017.02.006 -



 下载:
下载: