-
青藏高原被誉为世界第三极,其冰雪含量仅次于南极和北极[1],在整个亚洲大陆的大气环流和水循环中起着重要作用[2 − 3]. 尽管青藏高原是全球最干净的地区之一[4],但其大气环境也受到来自远处的污染物输送和本地排放的影响[5 − 6],其中包括化石燃料和生物质燃烧产生的黑碳(black carbon,BC)[7 − 8]. BC在大气中能有效吸收几乎整个太阳光谱的太阳辐射,对大气层产生显著的辐射效应,从而影响气候变化[9]. 一旦BC沉降到冰雪表面,其强吸光性会显著降低雪冰表面反照率,导致雪冰表面更多地吸收太阳辐射[10]. 此外,BC经过老化后其吸光能力明显增强[11],进一步加速冰雪融化. 最新研究发现,青藏高原冰雪的快速退缩与冰雪中沉积的BC有关[12]. 由于青藏高原地处高寒缺能区域,牧区牧民经常使用牛羊粪作为生物质燃料取暖和烹饪. 该地区生物质能源消费在家庭能源消费中占比达41.4%,其中牦牛粪是重要来源之一[13].
目前,研究更多关注外来BC对青藏高原气候和生态环境的影响,对本地生物质燃料燃烧排放BC的研究则相对较少. 例如,Liu等[14]的研究表明,生物质燃烧对青藏高原东南缘BC质量浓度贡献占57%,主要来自印度东北部和缅甸北部输送;Yang等[15]使用区域大气化学模型WRF-Chem发现,南亚居民排放对青藏高原BC浓度贡献最大,传输过程中夏季为25.8%,冬季为44.8%. 尽管已有研究团队对当地居民燃烧牦牛粪排放的气溶胶进行了研究,但对青藏高原牦牛粪燃烧产生BC颗粒物的化学特性了解仍较少,特别是对其老化后的化学特征和混合状态的变化. 例如,Chen等[6]的研究表明,牦牛粪排放的气溶胶中有机碳(OC)和BC分别占55.2%和3.63%,所研究气溶胶的OC/BC比值为16.3±4.4,接近环境气溶胶OC/BC值(季风期为21.7,非季风期为23.1),进一步表明当地来源对青藏高原大气的影响.
研究当地居民燃烧牦牛粪排放BC颗粒物的化学特性对准确评估青藏高原的污染源具有重要作用. 本研究通过燃烧模拟实验,利用氧化流动反应器(potential aerosol mass,PAM)模拟牦牛粪燃烧产生的BC气溶胶在大气中的老化过程,然后采用单颗粒气溶胶质谱仪(single-particle aerosol mass spectrometer,SPAMS)对BC颗粒物的化学特征以及混合状态进行表征.
-
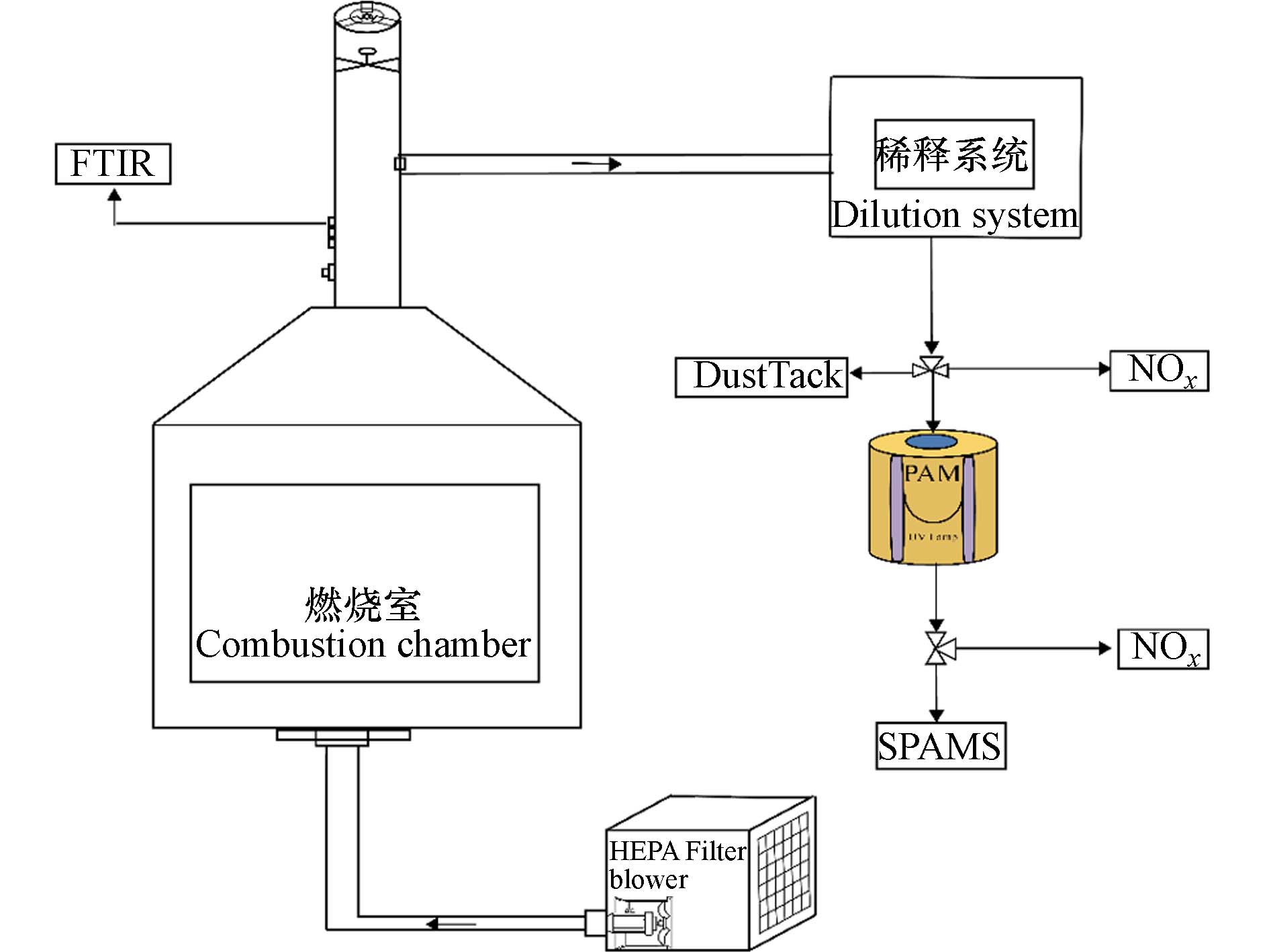
牦牛粪燃烧模拟实验在一个自制的燃烧模拟腔内进行. 该燃烧室为一个长宽高分别为1.8 m、1.8 m和2.2 m,容积为8 m3的空腔容器. 燃烧室壁采用3 mm厚的钝化铝材料,能够承受高温燃烧. 燃烧室内配备热电偶、热风速计和空气净化系统. 安装在腔室上方的稀释采样器(18 Model,Baldwin Environmental Inc.,Reno,NV,USA),能够将高浓度的采样烟气进行一定倍数的稀释,为了减少烟气颗粒物的损失,整个采样器的金属组件均采用不锈钢. 在实验室筛选粒径为0.5—10 µm的聚苯乙烯乳胶小球(polystyrene latex spheres,PSL),并以100 L·min−1的流速通过稀释通道采样器,结果显示0.5—5 µm的PSL的捕捉率是100%,考虑到颗粒物从排放源到进入稀释采样器之前在采样管路中的损失,得到稀释通道的PM2.5损失<5%. Tian等[16]报道了该燃烧室的详细结构和性能评估.
牦牛粪样品采集自西藏自治区那曲市周边的典型高原牧区,分为两组样品,分别来自海拔
4508.9 m和5054.7 m的地区. 每次取约5 g等分样品,使用已经燃烧通红的蜂窝煤引燃,整个燃烧过程约 10—20 min. 如图1所示,牦牛粪燃烧产生的烟气被稀释采样器稀释后进入PAM反应器,以模拟牧民燃烧牦牛粪产生的BC颗粒物在大气中的老化过程. 最后,利用SPAMS对BC颗粒物在老化前后的化学组分和混合状态的演变特征进行表征,同时探究BC颗粒物上二次组分的形成潜力. -
PAM是一种氧化流动反应器,可提供高度氧化的环境,模拟一天至几天的大气氧化过程,用于模拟燃烧源排放烟气在大气中的老化过程. 该反应器长 46 cm ×宽 22 cm的铝制圆柱体,内部容积为 13.3 L . PAM 反应器内置紫外灯,发射 185 nm 和 254 nm 的光子,以产生大量的O3和·OH,产生氧化剂的反应如下:
该反应器在不同的流速下对不同粒径具有不同的传输效率,在流速为 8.5 L·min−1时,对于迁移直径>150 nm 的颗粒,传输效率超过 80%,但对于 50 nm 的颗粒,传输效率低至 40%;在流速为 9.75 L·min−1时,20 nm 颗粒的颗粒损失为 60%,约 50 nm 颗粒的颗粒损失为约 25%,对于>100 nm的颗粒,其损失不超过10%[17]. 反应器内的停留时间理想地是PAM体积除以体积流量,其范围为 10 L·min−1流量的约 80 s到 1 L·min−1流量的约 800 s,但由于PAM的抛物线轮廓、入口区域的射流和再循环以及大气成分的“黏性”会使得停留时间变得复杂,在本课题组以往的研究中表明CO2在9 L·min−1流速下停留时间约为90 s[17].
本研究通过调节PAM内置紫外灯的电压,控制不同水平的·OH自由基的暴露水平,模拟牦牛粪燃烧排放的BC颗粒物在大气中的老化过程. 在本研究中,选择3个模拟的大气老化程度:0 d、3 d和7 d,其中0 d表示未经过模拟老化,根据大气中平均·OH自由基浓度1.5× 106 molecules·cm−3[17], 3 d和7 d分别对应的·OH自由基暴露值(OHexp)为4.6 × 1011 molecules·s·cm−3和8.8 × 1011 molecules·s·cm−3. 值得注意的是,真实大气中的·OH自由基浓度在不同的环境中可能会存在1个数量级的变化,因此在本研究中通过PAM暴露换算的老化天数具有不可忽略的不确定性. Cao 等[17]在其研究中详细介绍了紫外灯电压对应的老化程度.
-
SPAMS(型号 0515,广州禾信仪器股份有限公司,中国)可实时在线快速检测单颗粒气溶胶,获得空气动力学尺寸为 0.2 μm至 2.0 μm的单颗粒物,可测量的化学成分的m/z为± 250 amu. SPAMS 的流速为 75 mL·min−1,其工作原理已在其他文章中详细阐述[18]. 简而言之,气溶胶颗粒进入SPAMS后,会立即聚焦并通过空气动力学透镜加速到不同的速度. 通过透镜加速后,气溶胶颗粒的速度由其空气动力学直径决定. 然后,通过在分级区域使用两个固定距离(6 cm)的连续钕/钇铝石榴石(Nd: YAG))二极管激光器(532 nm)获得气溶胶颗粒的空气动力学直径. 使用 266 nm 紫外激光束通过解吸/电离过程检测气溶胶颗粒的化学成分. 产生的正离子和负离子碎片都用真空空气动力学直径(dva)记录. 在 SPAMS 分析之前,对正负离子碎片的空气动力学直径和质荷比进行了校准和优化[19]. 在采样期间,清洁了关键孔口中的污染物,并将入口压力控制在(2.40 ± 0.05) Torr 以内.
本研究使用基于MATLAB(版本,2014b,www.mathworks.com)运行的COCO V1.4 软件包分析SPAMS 获得的数据,应用基于自适应共振理论的神经网络算法(ART-2a),根据单个粒子质谱中离子峰的存在和强度将单个粒子聚类成单独的组[20]. 在此算法中,采用的相似度、学习率和迭代次数分别为0.60、0.05 和 20. 当质谱特征中同时出现12C+、24C2+、36C3+、48C4+和60C5+或12C−、24C2−、36C3−、48C4−和60C5−特征峰时,被筛选为BC颗粒.
-
图2为牦牛粪燃烧排放的BC颗粒物在不同模拟老化程度下的平均质谱特征. 为了方便描述,将牦牛粪燃烧排放的含BC颗粒物简称为BC颗粒. 结果显示,不同老化程度的BC颗粒在正质谱图中主要包含了39K+和有机碳(OC)等离子碎片,并且OC的相对峰面积最显著. OC质谱峰主要包括m/z 27C2H3+/CNH+、37C3H+、43C2H3O+、50C4H2+、51C4H3+和63C5H3+,它们相对峰面积总和超过40%(0 d为42.8%,3 d为44%,7 d为42.1%). Zhang等[21]研究发现,牦牛粪燃烧产生的有机物贡献了亚微米气溶胶质量的95%,因此本文中OC相对峰面积最显著的主要原因可能是牦牛粪燃烧过程中产生的大量有机物附着在BC颗粒上. 39K+是生物质燃烧颗粒物的标记物[22].
在负质谱图中,97HSO4−是主要的离子峰,表明BC颗粒混合了大量硫酸盐. 这可能是由于在燃烧过程中,钾相对于硫和氯以化学计量或超化学计量的量(eK/(eCl+2eS)≥1,其中eK、eCl和eS为K、Cl和S的元素总浓度),导致所有硫(以SO2形式释放)在硫酸盐化平衡的影响下被氧化为硫酸盐,以及硫酸盐的平衡蒸汽压更高更容易在硫酸盐颗粒相形成中过饱和沉淀[23]. 此外,负质谱图中还有常见的生物质燃烧颗粒26CN−和42CNO−质谱特征,其来源于牦牛粪中游离脂质或生物聚合物在不完全燃烧过程中的热分解[24 − 25].
未经模拟老化的BC颗粒中,46NO2−和62NO3−的相对峰面积较低,分别占负离子相对面积的2.7%和1.3%,表明新鲜排放的BC颗粒中硝酸盐含量较少. 经过3 d和7 d模拟大气老化后,相较于未经模拟老化, 62NO3−的相对峰面积明显增加,分别增加了7.3%和11.5%. 这主要是由于在PAM反应器中·OH自由基浓度的增加,使得更多的NO2通过非均相反应与·OH自由基作用(具体反应如:
NO2+⋅OH→HNO3 ),从而生成更多的气态HNO3,附着在BC颗粒物表面后向颗粒态转移[26]. 而在模拟大气老化后的BC颗粒物中,97HSO4−的相对峰面积有所减少,这可能是由其他物质的增多带来的变化.为了更清楚地了解牦牛粪BC颗粒物上有机化学组分的变化情况,通过对模拟老化后的BC颗粒相对峰面积与未经老化的BC颗粒相对峰面积进行差分分析,探索了不同老化程度的牦牛粪BC颗粒的差分质谱峰. 图3展示了牦牛粪BC模拟老化3 d和7 d与未经老化的差分质谱特征. 结果显示,在模拟老化3 d和模拟老化7 d的BC颗粒中,OC的相对峰面积变化并不明显. 例如:3 d和7 d中43C2H3O+分别增加了0.95%和0.72%; 37C3H+分别减少了0.75%和0.93%;63C5H3+分别减少了0.21%和0.34%. 这可能是由于老化过程中有机物形成和降解的竞争,导致新鲜和老化的BC颗粒物中有机物的含量没有显著变化[27].
-
基于以下几种原则对不同BC颗粒进行了分类:(1)当质谱特征中只出现12C+、24C2+、36C3+、48C4+和60C5+或12C−、24C2−、36C3−、48C4−和60C5−特征峰时,将其命名为纯BC;(2)当OC(27C2H3+/CNH+、37C3H+、43C2H3O+、50C4H2+、51C4H3+和63C5H3+)的相对峰面积之和与正离子区的Cn+(n=1,2,3……)相对峰面积相当,并且97HSO4−的相对峰面积大于50%时,将其命名为BCOCS. (3)当BC颗粒只含有46NO2−和62NO3−特征峰,且相对峰面积之和大于70%时,将其命名为BCN;(4)不符合以上3个命名原则的BC颗粒则被命名为Other[28]. 通过计算所选离子碎片46NO2−、18NH4+、62NO3−、97HSO4−、43C2H3O+和89HC2O4−对每种颗粒类型的贡献,研究了牦牛粪燃烧产生BC颗粒的混合状态.
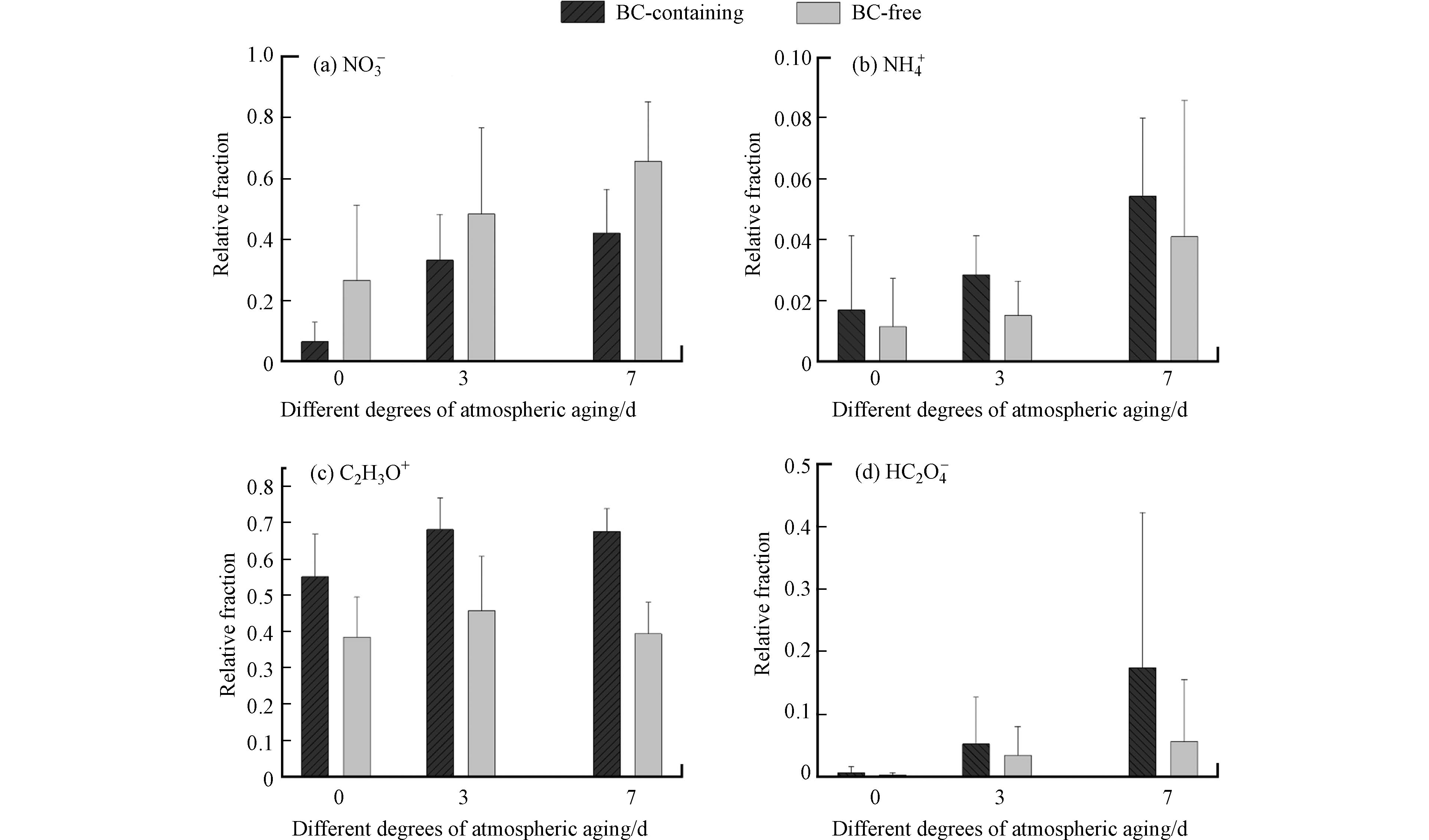
图4为不同模拟老化程度下牦牛粪燃烧排放含BC颗粒的混合态特征. 其中色块的颜色深浅代表含有对应所选离子碎片与该类型颗粒物总数的比值,能够在一定程度上反映该类型颗粒物与所选定离子的混合程度. 结果显示,纯BC颗粒类型不与各离子碎片混合,说明该类型颗粒为外混BC. 在3种模拟老化条件下,46NO2−均与各BC颗粒类型混合,在BCN中46NO2−的占比均超过70%,Other类型中只有模拟老化0 d和7 d超过60%,而BCOCS中3种模拟老化情况下46NO2−的占比均不超过15%. 在未经模拟老化条件下,18NH+不与任何BC颗粒类型混合(图4a);模拟大气老化3 d后,18NH+只混合在了BCN类型中(图4b),占该颗粒类型的14%;模拟大气老化7 d后(图4c),18NH+虽然与BCOCS和Other类型出现了一定的混合,但混合占比并不高(<3%),主要混合在了BCN中,混合占比为30.2%,这说明18NH+可能更容易在含氮的BC颗粒物上老化形成.
在未经模拟老化的情况下,BCN、BCOCS和Other中含分别含有10%、49%和14%的43C2H3O+离子峰. 在模拟老化3 d后,BCN中含有43C2H3O+离子峰的颗粒增加了18.5%,BCOCS中增加了22.6%,Other中增加了8.7%. 43C2H3O+作为一种二次有机气溶胶的指示物[29],在模拟老化7 d后,含有43C2H3O+离子碎片的BC颗粒的数量分数明显增多,BCN、BCOCS和Other颗粒类型分别增加了18%、24%和8%.
89HC2O4−是另一种二次有机气溶胶指示物[30],在未经模拟老化的情况下并没有发现草酸盐离子峰. 在模拟老化3 d后,BCN中含有21.4%的89HC2O4−离子峰,BCOCS中含有2.2%的89HC2O4−离子峰,Other中不含有89HC2O4−离子峰. 在模拟老化7 d中,相较于模拟老化3 d,BCN中含有89HC2O4−离子峰的颗粒下降了13.9%,BCOCS中则增加了4.9%,Other中增加了29.3%. 模拟老化7 d中BCN的数量明显多于3 d,而89HC2O4−的数量增长并不明显,这说明62NO3−的增长速率要明显高于89HC2O4−的速率,因此在模拟老化7 d中89HC2O4−在BCN中的占比明显降低.
97HSO4−大量混合在BCOCS中,并且其占比在不同大气老化程度中都达到99%,导致硫酸盐大量混合在含OC的BC颗粒上. 主要原因可能是气态硫酸和有机蒸汽相互作用,加速了硫酸-有机颗粒物的形成. 牟等人[31]通过SPAMS观测上海秋季气溶胶时也发现硫酸盐在含OC的BC颗粒中有强烈的信号峰. 在未经模拟老化的情况下,BCN中含有45%的97HSO4−离子峰,Other中含有52%的97HSO4−离子峰. 模拟老化3 d后,BCN中含有97HSO4−离子峰的颗粒增加了5%,而Other中则不变. 经过模拟老化7 d后,Other的占比增加到99.0%,而BCN的占比与模拟老化3 d相比则基本不变(<1%),说明中等时间的老化并不能明显增多BC颗粒上的硫酸盐.
62NO3−大量混合在了BCN中,其占比在不同大气老化程度中都超过了98%,这主要与其命名原则有关. 在模拟老化3 d后,BCOCS中仅含有2%的62NO3−离子峰,而Other中含有33%的62NO3−离子峰. 在模拟老化7 d后,BCOCS中增加了35%,而Other则100%的含有62NO3−离子峰. 这与气态HNO3的沉积和NO2在颗粒物表面的非均相反应增加有关.
-
本研究使用了18NH4+、62NO3−、43C2H3O+和89HC2O4−离子碎片在BC颗粒中的占比来表征不同老化条件下二次组分的形成机制[32 − 33]. 该占比的计算方式是将包含某二次组分的BC颗粒数量与整体BC颗粒数量进行比较,以得到相应的占比. 非BC颗粒中二次组分的占比计算方式与之类似.
如图5所示,含62NO3−的BC颗粒的占比与老化程度呈正相关,说明62NO3−的形成与·OH自由基的浓度有关,且与它们呈正相关. 在PAM反应器中,颗粒物中HNO3可能来源于NO2在·OH自由基的暴露下光氧化产生的气态HNO3的沉积,以及NO2在颗粒物表面的非均相反应[26]. 这一结果与外场观测的研究结果一致,62NO3−的数量分数与氧化剂(Ox=O3+NO2)的浓度呈正相关[32]. 含有18NH4+的BC颗粒的数量分数随老化程度的增强而增加(图5b),这可能是由于老化程度增加促进了HNO3的形成,且HNO3与燃烧产生的NH3(实验过程中FTIR测得烟气中NH3浓度为(417±68) μg·m-³)进一步中和导致了18NH4+的数量分数增加.
43C2H3O+和89HC2O4−是两种重要的二次有机物指示剂,它们的增长变化趋势存在一定差异(图5c、d). 在BC颗粒上,43C2H3O+的数量分数并没有随着老化程度的增强而持续增长,而是趋于平稳,即3 d老化和7 d老化之间没有明显差别;但89HC2O4−的数量分数随着老化程度的增强逐渐变大. 研究表明,有机物被·OH自由基氧化后,会经过官能化(functionalization)和碎片化(fragmentation)两个阶段,官能化会在氧化早期增加有机物的质量,而碎片化在氧化后期能够减少有机物的质量[26]. 因此,老化后43C2H3O+数量分数变化不明显的原因可能是其官能化和碎片化之间形成了平衡. 相比之下,89HC2O4−的数量分数随着老化程度的增强逐渐增大. 这可能是由于牦牛粪燃烧产生大量的挥发性有机物(VOCs)[34],而89HC2O4−的主要生成途径为乙二醛和乙醛酸的光化学氧化,高浓度的氧化剂会导致更多的VOCs 产生二羰基和醛类物质(如乙二醛)[32,35-37],从而增加了89HC2O4−的前体物浓度,因此随着PAM中·OH自由基浓度的增加,89HC2O4−的数量分数也逐渐变大.
最后,本研究还对比了非BC颗粒上二次组分形成情况,以研究BC在二次组分形成过程中的作用(图5). 结果显示,二次组分18NH4+、43C2H3O+和89HC2O4−在非BC颗粒和BC颗粒中数量分数的增长变化趋势基本一致. BC颗粒中这些二次组分的数量分数明显高于非BC颗粒,这说明BC在促进BC包裹物中二次组分形成具有一定作用. 研究发现BC可以作为光活性物质和吸收基质,导致这些二次组分富集在BC颗粒上[38 − 39]. 最近的研究还发现BC在与BC包裹物上硫酸盐的演化中具有重要作用[40]. 值得注意的是,尽管BC颗粒与非BC颗粒上62NO3−的增长趋势趋同,但不论是新鲜排放还是模拟老化后,非BC颗粒中62NO3−的数量分数都明显高于BC颗粒. 这可能是由于在燃烧过程中非BC颗粒的数量明显高于BC颗粒,导致PAM反应器中气态HNO3的沉积和NO2在颗粒物表面的非均相反应更多集中在非BC颗粒上.
-
为了更准确地解释青藏高原地区BC颗粒对气候和环境影响,在区域气溶胶气候模型中有必要考虑BC颗粒的光化学反应,因为化学组分的变化对于评估BC颗粒的吸湿性和光学特性至关重要. 因此,本研究通过SPAMS实时在线分析了不同模拟老化程度下牦牛粪燃烧排放BC颗粒物的化学组成、混合状态和老化机制,在实验室条件下证明了BC能够促进一些二次组分在BC颗粒上的富集,具体结论如下:
(1)不同老化程度下牦牛粪燃烧排放的含BC颗粒物均主要含有OC、46NO2–、62NO3−和97HSO4−,OC在正离子相对峰面积中占主导,其在0 d、3 d和7 d的占比分别为42.8%、44%和42.1%,而97HSO4−在负离子相对峰面积中占主导,其中0 d、3 d和7 d的占比分别为72.4%、64.6%和64.6%,并且随着老化程度的增加62NO3−的相对峰面积显著增加,这与气态HNO3的沉积和NO2在颗粒物表面的非均相反应增加有关.
(2)在含BC颗粒中,43C2H3O+在3种主要类型颗粒物中的混合占比随着老化程度的增强而增加;97HSO4−在所有颗粒物中均占主导地位,且老化程度的影响较小;而62NO3−主要存在于BCN颗粒中,但是在BCOCS和Other颗粒物中的混合程度随老化程度的增强而增加.
(3)二次组62NO3−、18NH4+和89HC2O4−的数量百分比与老化时间均呈正相关关系,即老化程度越高颗粒物中它们的相对含量越高;而随着老化程度的增加,43C2H3O+的数量占比呈现先上升后趋于稳定的趋势.
牦牛粪燃烧排放黑碳的化学老化过程
Chemical aging process of black carbon emissions from yak dung combustion
-
摘要: 当前,对青藏高原牦牛粪燃烧排放的含黑碳(BC)颗粒物在老化过程中的演变机制了解还不足. 为深入研究这一问题,本文采用氧化流动反应器模拟牦牛粪燃烧排放BC颗粒物的不同老化程度,并利用单颗粒气溶胶质谱仪对老化前后BC颗粒物的化学组成、混合状态、老化机制等进行了表征. 结果显示,在不同程度的老化模拟过程中,牦牛粪燃烧排放的BC颗粒物均呈现显著的有机碳(OC)、硫酸盐(HSO4−)和硝酸盐(NO3−)离子峰特征. 老化前,有机碳(其特征峰主要为m/z = 27C2H3+/CNH+、37C3H+、43C2H3O+、50C4H2+、51C4H3+和63C5H3+)在正离子相对峰面积中占比为42.8%,而硫酸盐在负离子相对峰面积中占比高达72.4%. 基于自适应共振神经网络算法(ART-2a),将BC颗粒物分为4类:BC、BCN、BCOCS和Other. 硫酸盐与BCOCS颗粒类型高度混合(99%),而硝酸盐与BCN颗粒类型高度混合(98%). 此外,随着老化程度的增加,二次组分(如43C2H3O和62NO3−)在各颗粒类型中的混合程度也越高. 硝酸盐、草酸盐(89HC2O4−)和铵盐(18NH4+)的数量百分比与老化程度呈显著的正相关关系,表明在BC颗粒物老化的过程中它们的贡献会增加. 本研究有助于更加全面地理解青藏高原大气中BC颗粒物的行为,为该区域大气环境保护提供科学依据.Abstract: Currently, the evolution mechanism of black carbon (BC) particles emitted from yak dung combustion on the Qinghai-Tibet Plateau during the aging process remains poorly understood. In order to further study these issues, an oxidation flow reactor was used to simulate various aging degrees of BC particles emitted from yak dung combustion. The chemical compositions, mixing states, and aging mechanism of BC particles before and after aging were characterized by a single-particle aerosol mass spectrometer. The results showed that during the simulation of different aging degrees, the BC particles emitted from yak dung combustion exhibited distinctive peak characteristics of organic carbon (OC), sulfate (HSO4−), and nitrate (NO3−). Prior to aging, organic carbon (with characteristic peaks at m/z = 27C2H3+/CNH+37C3H+,43C2H3O+,50C4H2+,51C4H3+, and 63C5H3+) accounted for 42.8% of the relative peak area of positive ions, while sulfate accounted for 72.4% of the relative peak area of negative ions. Based on the adaptive resonance neural network algorithm (ART-2a), BC particles are divided into four categories: BC, BCN, BCOCS, and Other. Sulfates exhibited high mixing with the BCOCS particle type (99%), whereas nitrates showed high mixing with the BCN particle type (98%). Furthermore, as the aging degree increased, secondary components (such as 43C2H3O+ and 62NO3−) became more prevalent in each particle type. The percentages of nitrate, oxalate (89HC2O4−), and ammonium salt (18NH4+) positively correlated with the degree of aging, indicating an increased contribution during the aging of BC particles. This study contributes to a more comprehensive understanding of the behavior of BC particles in the atmosphere of the Qinghai-Tibet Plateau, providing a scientific basis for the protection of the atmospheric environment in this region.
-
Key words:
- Qinghai-Tibet Plateau /
- black carbon /
- chemical composition /
- mixing state /
- aging simulation
-
青藏高原被誉为世界第三极,其冰雪含量仅次于南极和北极[1],在整个亚洲大陆的大气环流和水循环中起着重要作用[2 − 3]. 尽管青藏高原是全球最干净的地区之一[4],但其大气环境也受到来自远处的污染物输送和本地排放的影响[5 − 6],其中包括化石燃料和生物质燃烧产生的黑碳(black carbon,BC)[7 − 8]. BC在大气中能有效吸收几乎整个太阳光谱的太阳辐射,对大气层产生显著的辐射效应,从而影响气候变化[9]. 一旦BC沉降到冰雪表面,其强吸光性会显著降低雪冰表面反照率,导致雪冰表面更多地吸收太阳辐射[10]. 此外,BC经过老化后其吸光能力明显增强[11],进一步加速冰雪融化. 最新研究发现,青藏高原冰雪的快速退缩与冰雪中沉积的BC有关[12]. 由于青藏高原地处高寒缺能区域,牧区牧民经常使用牛羊粪作为生物质燃料取暖和烹饪. 该地区生物质能源消费在家庭能源消费中占比达41.4%,其中牦牛粪是重要来源之一[13].
目前,研究更多关注外来BC对青藏高原气候和生态环境的影响,对本地生物质燃料燃烧排放BC的研究则相对较少. 例如,Liu等[14]的研究表明,生物质燃烧对青藏高原东南缘BC质量浓度贡献占57%,主要来自印度东北部和缅甸北部输送;Yang等[15]使用区域大气化学模型WRF-Chem发现,南亚居民排放对青藏高原BC浓度贡献最大,传输过程中夏季为25.8%,冬季为44.8%. 尽管已有研究团队对当地居民燃烧牦牛粪排放的气溶胶进行了研究,但对青藏高原牦牛粪燃烧产生BC颗粒物的化学特性了解仍较少,特别是对其老化后的化学特征和混合状态的变化. 例如,Chen等[6]的研究表明,牦牛粪排放的气溶胶中有机碳(OC)和BC分别占55.2%和3.63%,所研究气溶胶的OC/BC比值为16.3±4.4,接近环境气溶胶OC/BC值(季风期为21.7,非季风期为23.1),进一步表明当地来源对青藏高原大气的影响.
研究当地居民燃烧牦牛粪排放BC颗粒物的化学特性对准确评估青藏高原的污染源具有重要作用. 本研究通过燃烧模拟实验,利用氧化流动反应器(potential aerosol mass,PAM)模拟牦牛粪燃烧产生的BC气溶胶在大气中的老化过程,然后采用单颗粒气溶胶质谱仪(single-particle aerosol mass spectrometer,SPAMS)对BC颗粒物的化学特征以及混合状态进行表征.
1. 实验方法(Experimental methods)
1.1 牦牛粪燃烧排放模拟实验
牦牛粪燃烧模拟实验在一个自制的燃烧模拟腔内进行. 该燃烧室为一个长宽高分别为1.8 m、1.8 m和2.2 m,容积为8 m3的空腔容器. 燃烧室壁采用3 mm厚的钝化铝材料,能够承受高温燃烧. 燃烧室内配备热电偶、热风速计和空气净化系统. 安装在腔室上方的稀释采样器(18 Model,Baldwin Environmental Inc.,Reno,NV,USA),能够将高浓度的采样烟气进行一定倍数的稀释,为了减少烟气颗粒物的损失,整个采样器的金属组件均采用不锈钢. 在实验室筛选粒径为0.5—10 µm的聚苯乙烯乳胶小球(polystyrene latex spheres,PSL),并以100 L·min−1的流速通过稀释通道采样器,结果显示0.5—5 µm的PSL的捕捉率是100%,考虑到颗粒物从排放源到进入稀释采样器之前在采样管路中的损失,得到稀释通道的PM2.5损失<5%. Tian等[16]报道了该燃烧室的详细结构和性能评估.
牦牛粪样品采集自西藏自治区那曲市周边的典型高原牧区,分为两组样品,分别来自海拔
4508.9 m和5054.7 m的地区. 每次取约5 g等分样品,使用已经燃烧通红的蜂窝煤引燃,整个燃烧过程约 10—20 min. 如图1所示,牦牛粪燃烧产生的烟气被稀释采样器稀释后进入PAM反应器,以模拟牧民燃烧牦牛粪产生的BC颗粒物在大气中的老化过程. 最后,利用SPAMS对BC颗粒物在老化前后的化学组分和混合状态的演变特征进行表征,同时探究BC颗粒物上二次组分的形成潜力.1.2 PAM反应器
PAM是一种氧化流动反应器,可提供高度氧化的环境,模拟一天至几天的大气氧化过程,用于模拟燃烧源排放烟气在大气中的老化过程. 该反应器长 46 cm ×宽 22 cm的铝制圆柱体,内部容积为 13.3 L . PAM 反应器内置紫外灯,发射 185 nm 和 254 nm 的光子,以产生大量的O3和·OH,产生氧化剂的反应如下:
H2O +hv(185 nm)→⋅OH + H (1) O2+hv(185 nm)→2O(3P) (2) O2+O(3P)→O3 (3) O3+hv(254 nm )→O2+ O(1D) (4) O(1D) +H2O → 2OH (5) H +O2→ HO2 (6) 该反应器在不同的流速下对不同粒径具有不同的传输效率,在流速为 8.5 L·min−1时,对于迁移直径>150 nm 的颗粒,传输效率超过 80%,但对于 50 nm 的颗粒,传输效率低至 40%;在流速为 9.75 L·min−1时,20 nm 颗粒的颗粒损失为 60%,约 50 nm 颗粒的颗粒损失为约 25%,对于>100 nm的颗粒,其损失不超过10%[17]. 反应器内的停留时间理想地是PAM体积除以体积流量,其范围为 10 L·min−1流量的约 80 s到 1 L·min−1流量的约 800 s,但由于PAM的抛物线轮廓、入口区域的射流和再循环以及大气成分的“黏性”会使得停留时间变得复杂,在本课题组以往的研究中表明CO2在9 L·min−1流速下停留时间约为90 s[17].
本研究通过调节PAM内置紫外灯的电压,控制不同水平的·OH自由基的暴露水平,模拟牦牛粪燃烧排放的BC颗粒物在大气中的老化过程. 在本研究中,选择3个模拟的大气老化程度:0 d、3 d和7 d,其中0 d表示未经过模拟老化,根据大气中平均·OH自由基浓度1.5× 106 molecules·cm−3[17], 3 d和7 d分别对应的·OH自由基暴露值(OHexp)为4.6 × 1011 molecules·s·cm−3和8.8 × 1011 molecules·s·cm−3. 值得注意的是,真实大气中的·OH自由基浓度在不同的环境中可能会存在1个数量级的变化,因此在本研究中通过PAM暴露换算的老化天数具有不可忽略的不确定性. Cao 等[17]在其研究中详细介绍了紫外灯电压对应的老化程度.
1.3 单颗粒气溶胶质谱仪和数据分析
SPAMS(型号 0515,广州禾信仪器股份有限公司,中国)可实时在线快速检测单颗粒气溶胶,获得空气动力学尺寸为 0.2 μm至 2.0 μm的单颗粒物,可测量的化学成分的m/z为± 250 amu. SPAMS 的流速为 75 mL·min−1,其工作原理已在其他文章中详细阐述[18]. 简而言之,气溶胶颗粒进入SPAMS后,会立即聚焦并通过空气动力学透镜加速到不同的速度. 通过透镜加速后,气溶胶颗粒的速度由其空气动力学直径决定. 然后,通过在分级区域使用两个固定距离(6 cm)的连续钕/钇铝石榴石(Nd: YAG))二极管激光器(532 nm)获得气溶胶颗粒的空气动力学直径. 使用 266 nm 紫外激光束通过解吸/电离过程检测气溶胶颗粒的化学成分. 产生的正离子和负离子碎片都用真空空气动力学直径(dva)记录. 在 SPAMS 分析之前,对正负离子碎片的空气动力学直径和质荷比进行了校准和优化[19]. 在采样期间,清洁了关键孔口中的污染物,并将入口压力控制在(2.40 ± 0.05) Torr 以内.
本研究使用基于MATLAB(版本,2014b,www.mathworks.com)运行的COCO V1.4 软件包分析SPAMS 获得的数据,应用基于自适应共振理论的神经网络算法(ART-2a),根据单个粒子质谱中离子峰的存在和强度将单个粒子聚类成单独的组[20]. 在此算法中,采用的相似度、学习率和迭代次数分别为0.60、0.05 和 20. 当质谱特征中同时出现12C+、24C2+、36C3+、48C4+和60C5+或12C−、24C2−、36C3−、48C4−和60C5−特征峰时,被筛选为BC颗粒.
2. 结果与讨论(Results and discussion)
2.1 不同老化程度BC颗粒质谱特征
图2为牦牛粪燃烧排放的BC颗粒物在不同模拟老化程度下的平均质谱特征. 为了方便描述,将牦牛粪燃烧排放的含BC颗粒物简称为BC颗粒. 结果显示,不同老化程度的BC颗粒在正质谱图中主要包含了39K+和有机碳(OC)等离子碎片,并且OC的相对峰面积最显著. OC质谱峰主要包括m/z 27C2H3+/CNH+、37C3H+、43C2H3O+、50C4H2+、51C4H3+和63C5H3+,它们相对峰面积总和超过40%(0 d为42.8%,3 d为44%,7 d为42.1%). Zhang等[21]研究发现,牦牛粪燃烧产生的有机物贡献了亚微米气溶胶质量的95%,因此本文中OC相对峰面积最显著的主要原因可能是牦牛粪燃烧过程中产生的大量有机物附着在BC颗粒上. 39K+是生物质燃烧颗粒物的标记物[22].
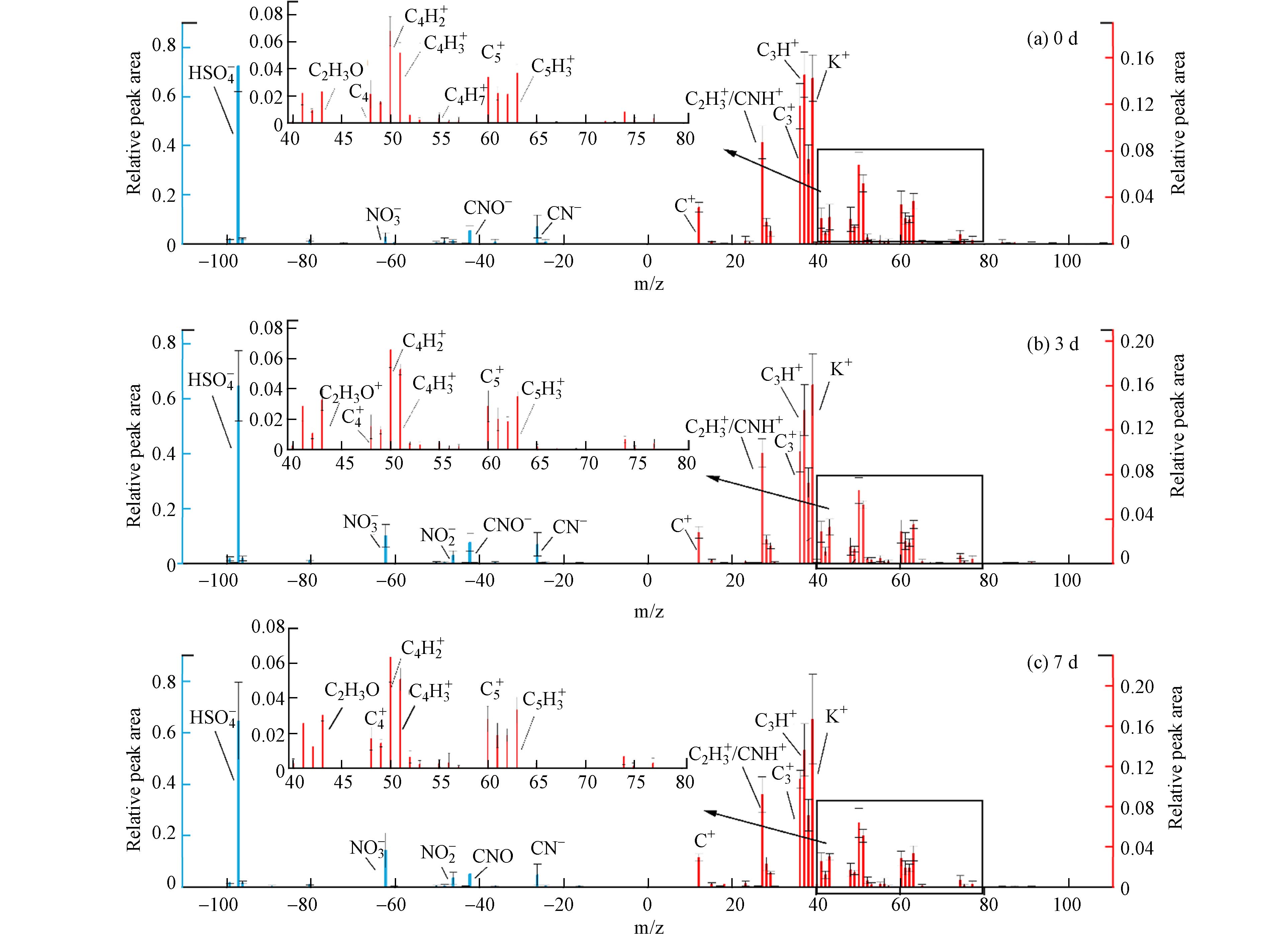 图 2 不同老化程度下牦牛粪BC的平均质谱特征Figure 2. Average mass spectrometry of the emitted BC-containing particles from BC yak dung combustion at different aging degrees ,including 0 day (a), 3day (b) and 7 day (c) respectively(右侧为正质谱特征,左侧为负质谱特征)(Red refer to the positive mass spectrometry, blue refer to negative mass spectrometry)
图 2 不同老化程度下牦牛粪BC的平均质谱特征Figure 2. Average mass spectrometry of the emitted BC-containing particles from BC yak dung combustion at different aging degrees ,including 0 day (a), 3day (b) and 7 day (c) respectively(右侧为正质谱特征,左侧为负质谱特征)(Red refer to the positive mass spectrometry, blue refer to negative mass spectrometry)在负质谱图中,97HSO4−是主要的离子峰,表明BC颗粒混合了大量硫酸盐. 这可能是由于在燃烧过程中,钾相对于硫和氯以化学计量或超化学计量的量(eK/(eCl+2eS)≥1,其中eK、eCl和eS为K、Cl和S的元素总浓度),导致所有硫(以SO2形式释放)在硫酸盐化平衡的影响下被氧化为硫酸盐,以及硫酸盐的平衡蒸汽压更高更容易在硫酸盐颗粒相形成中过饱和沉淀[23]. 此外,负质谱图中还有常见的生物质燃烧颗粒26CN−和42CNO−质谱特征,其来源于牦牛粪中游离脂质或生物聚合物在不完全燃烧过程中的热分解[24 − 25].
未经模拟老化的BC颗粒中,46NO2−和62NO3−的相对峰面积较低,分别占负离子相对面积的2.7%和1.3%,表明新鲜排放的BC颗粒中硝酸盐含量较少. 经过3 d和7 d模拟大气老化后,相较于未经模拟老化, 62NO3−的相对峰面积明显增加,分别增加了7.3%和11.5%. 这主要是由于在PAM反应器中·OH自由基浓度的增加,使得更多的NO2通过非均相反应与·OH自由基作用(具体反应如:
NO2+⋅OH→HNO3 为了更清楚地了解牦牛粪BC颗粒物上有机化学组分的变化情况,通过对模拟老化后的BC颗粒相对峰面积与未经老化的BC颗粒相对峰面积进行差分分析,探索了不同老化程度的牦牛粪BC颗粒的差分质谱峰. 图3展示了牦牛粪BC模拟老化3 d和7 d与未经老化的差分质谱特征. 结果显示,在模拟老化3 d和模拟老化7 d的BC颗粒中,OC的相对峰面积变化并不明显. 例如:3 d和7 d中43C2H3O+分别增加了0.95%和0.72%; 37C3H+分别减少了0.75%和0.93%;63C5H3+分别减少了0.21%和0.34%. 这可能是由于老化过程中有机物形成和降解的竞争,导致新鲜和老化的BC颗粒物中有机物的含量没有显著变化[27].
2.2 不同老化程度BC颗粒物的混合态特征
基于以下几种原则对不同BC颗粒进行了分类:(1)当质谱特征中只出现12C+、24C2+、36C3+、48C4+和60C5+或12C−、24C2−、36C3−、48C4−和60C5−特征峰时,将其命名为纯BC;(2)当OC(27C2H3+/CNH+、37C3H+、43C2H3O+、50C4H2+、51C4H3+和63C5H3+)的相对峰面积之和与正离子区的Cn+(n=1,2,3……)相对峰面积相当,并且97HSO4−的相对峰面积大于50%时,将其命名为BCOCS. (3)当BC颗粒只含有46NO2−和62NO3−特征峰,且相对峰面积之和大于70%时,将其命名为BCN;(4)不符合以上3个命名原则的BC颗粒则被命名为Other[28]. 通过计算所选离子碎片46NO2−、18NH4+、62NO3−、97HSO4−、43C2H3O+和89HC2O4−对每种颗粒类型的贡献,研究了牦牛粪燃烧产生BC颗粒的混合状态.
图4为不同模拟老化程度下牦牛粪燃烧排放含BC颗粒的混合态特征. 其中色块的颜色深浅代表含有对应所选离子碎片与该类型颗粒物总数的比值,能够在一定程度上反映该类型颗粒物与所选定离子的混合程度. 结果显示,纯BC颗粒类型不与各离子碎片混合,说明该类型颗粒为外混BC. 在3种模拟老化条件下,46NO2−均与各BC颗粒类型混合,在BCN中46NO2−的占比均超过70%,Other类型中只有模拟老化0 d和7 d超过60%,而BCOCS中3种模拟老化情况下46NO2−的占比均不超过15%. 在未经模拟老化条件下,18NH+不与任何BC颗粒类型混合(图4a);模拟大气老化3 d后,18NH+只混合在了BCN类型中(图4b),占该颗粒类型的14%;模拟大气老化7 d后(图4c),18NH+虽然与BCOCS和Other类型出现了一定的混合,但混合占比并不高(<3%),主要混合在了BCN中,混合占比为30.2%,这说明18NH+可能更容易在含氮的BC颗粒物上老化形成.
在未经模拟老化的情况下,BCN、BCOCS和Other中含分别含有10%、49%和14%的43C2H3O+离子峰. 在模拟老化3 d后,BCN中含有43C2H3O+离子峰的颗粒增加了18.5%,BCOCS中增加了22.6%,Other中增加了8.7%. 43C2H3O+作为一种二次有机气溶胶的指示物[29],在模拟老化7 d后,含有43C2H3O+离子碎片的BC颗粒的数量分数明显增多,BCN、BCOCS和Other颗粒类型分别增加了18%、24%和8%.
89HC2O4−是另一种二次有机气溶胶指示物[30],在未经模拟老化的情况下并没有发现草酸盐离子峰. 在模拟老化3 d后,BCN中含有21.4%的89HC2O4−离子峰,BCOCS中含有2.2%的89HC2O4−离子峰,Other中不含有89HC2O4−离子峰. 在模拟老化7 d中,相较于模拟老化3 d,BCN中含有89HC2O4−离子峰的颗粒下降了13.9%,BCOCS中则增加了4.9%,Other中增加了29.3%. 模拟老化7 d中BCN的数量明显多于3 d,而89HC2O4−的数量增长并不明显,这说明62NO3−的增长速率要明显高于89HC2O4−的速率,因此在模拟老化7 d中89HC2O4−在BCN中的占比明显降低.
97HSO4−大量混合在BCOCS中,并且其占比在不同大气老化程度中都达到99%,导致硫酸盐大量混合在含OC的BC颗粒上. 主要原因可能是气态硫酸和有机蒸汽相互作用,加速了硫酸-有机颗粒物的形成. 牟等人[31]通过SPAMS观测上海秋季气溶胶时也发现硫酸盐在含OC的BC颗粒中有强烈的信号峰. 在未经模拟老化的情况下,BCN中含有45%的97HSO4−离子峰,Other中含有52%的97HSO4−离子峰. 模拟老化3 d后,BCN中含有97HSO4−离子峰的颗粒增加了5%,而Other中则不变. 经过模拟老化7 d后,Other的占比增加到99.0%,而BCN的占比与模拟老化3 d相比则基本不变(<1%),说明中等时间的老化并不能明显增多BC颗粒上的硫酸盐.
62NO3−大量混合在了BCN中,其占比在不同大气老化程度中都超过了98%,这主要与其命名原则有关. 在模拟老化3 d后,BCOCS中仅含有2%的62NO3−离子峰,而Other中含有33%的62NO3−离子峰. 在模拟老化7 d后,BCOCS中增加了35%,而Other则100%的含有62NO3−离子峰. 这与气态HNO3的沉积和NO2在颗粒物表面的非均相反应增加有关.
2.3 老化模拟过程中BC颗粒物上二次组分的形成能力
本研究使用了18NH4+、62NO3−、43C2H3O+和89HC2O4−离子碎片在BC颗粒中的占比来表征不同老化条件下二次组分的形成机制[32 − 33]. 该占比的计算方式是将包含某二次组分的BC颗粒数量与整体BC颗粒数量进行比较,以得到相应的占比. 非BC颗粒中二次组分的占比计算方式与之类似.
如图5所示,含62NO3−的BC颗粒的占比与老化程度呈正相关,说明62NO3−的形成与·OH自由基的浓度有关,且与它们呈正相关. 在PAM反应器中,颗粒物中HNO3可能来源于NO2在·OH自由基的暴露下光氧化产生的气态HNO3的沉积,以及NO2在颗粒物表面的非均相反应[26]. 这一结果与外场观测的研究结果一致,62NO3−的数量分数与氧化剂(Ox=O3+NO2)的浓度呈正相关[32]. 含有18NH4+的BC颗粒的数量分数随老化程度的增强而增加(图5b),这可能是由于老化程度增加促进了HNO3的形成,且HNO3与燃烧产生的NH3(实验过程中FTIR测得烟气中NH3浓度为(417±68) μg·m-³)进一步中和导致了18NH4+的数量分数增加.
43C2H3O+和89HC2O4−是两种重要的二次有机物指示剂,它们的增长变化趋势存在一定差异(图5c、d). 在BC颗粒上,43C2H3O+的数量分数并没有随着老化程度的增强而持续增长,而是趋于平稳,即3 d老化和7 d老化之间没有明显差别;但89HC2O4−的数量分数随着老化程度的增强逐渐变大. 研究表明,有机物被·OH自由基氧化后,会经过官能化(functionalization)和碎片化(fragmentation)两个阶段,官能化会在氧化早期增加有机物的质量,而碎片化在氧化后期能够减少有机物的质量[26]. 因此,老化后43C2H3O+数量分数变化不明显的原因可能是其官能化和碎片化之间形成了平衡. 相比之下,89HC2O4−的数量分数随着老化程度的增强逐渐增大. 这可能是由于牦牛粪燃烧产生大量的挥发性有机物(VOCs)[34],而89HC2O4−的主要生成途径为乙二醛和乙醛酸的光化学氧化,高浓度的氧化剂会导致更多的VOCs 产生二羰基和醛类物质(如乙二醛)[32,35-37],从而增加了89HC2O4−的前体物浓度,因此随着PAM中·OH自由基浓度的增加,89HC2O4−的数量分数也逐渐变大.
最后,本研究还对比了非BC颗粒上二次组分形成情况,以研究BC在二次组分形成过程中的作用(图5). 结果显示,二次组分18NH4+、43C2H3O+和89HC2O4−在非BC颗粒和BC颗粒中数量分数的增长变化趋势基本一致. BC颗粒中这些二次组分的数量分数明显高于非BC颗粒,这说明BC在促进BC包裹物中二次组分形成具有一定作用. 研究发现BC可以作为光活性物质和吸收基质,导致这些二次组分富集在BC颗粒上[38 − 39]. 最近的研究还发现BC在与BC包裹物上硫酸盐的演化中具有重要作用[40]. 值得注意的是,尽管BC颗粒与非BC颗粒上62NO3−的增长趋势趋同,但不论是新鲜排放还是模拟老化后,非BC颗粒中62NO3−的数量分数都明显高于BC颗粒. 这可能是由于在燃烧过程中非BC颗粒的数量明显高于BC颗粒,导致PAM反应器中气态HNO3的沉积和NO2在颗粒物表面的非均相反应更多集中在非BC颗粒上.
3. 结论(Conclusions)
为了更准确地解释青藏高原地区BC颗粒对气候和环境影响,在区域气溶胶气候模型中有必要考虑BC颗粒的光化学反应,因为化学组分的变化对于评估BC颗粒的吸湿性和光学特性至关重要. 因此,本研究通过SPAMS实时在线分析了不同模拟老化程度下牦牛粪燃烧排放BC颗粒物的化学组成、混合状态和老化机制,在实验室条件下证明了BC能够促进一些二次组分在BC颗粒上的富集,具体结论如下:
(1)不同老化程度下牦牛粪燃烧排放的含BC颗粒物均主要含有OC、46NO2–、62NO3−和97HSO4−,OC在正离子相对峰面积中占主导,其在0 d、3 d和7 d的占比分别为42.8%、44%和42.1%,而97HSO4−在负离子相对峰面积中占主导,其中0 d、3 d和7 d的占比分别为72.4%、64.6%和64.6%,并且随着老化程度的增加62NO3−的相对峰面积显著增加,这与气态HNO3的沉积和NO2在颗粒物表面的非均相反应增加有关.
(2)在含BC颗粒中,43C2H3O+在3种主要类型颗粒物中的混合占比随着老化程度的增强而增加;97HSO4−在所有颗粒物中均占主导地位,且老化程度的影响较小;而62NO3−主要存在于BCN颗粒中,但是在BCOCS和Other颗粒物中的混合程度随老化程度的增强而增加.
(3)二次组62NO3−、18NH4+和89HC2O4−的数量百分比与老化时间均呈正相关关系,即老化程度越高颗粒物中它们的相对含量越高;而随着老化程度的增加,43C2H3O+的数量占比呈现先上升后趋于稳定的趋势.
-
-
[1] FARINOTTI D, HUSS M, FÜRST J J, et al. A consensus estimate for the ice thickness distribution of all glaciers on Earth[J]. Nature Geoscience, 2019, 12(3): 168-173. doi: 10.1038/s41561-019-0300-3 [2] JIN H J, HE R X, CHENG G D, et al. Changes in frozen ground in the Source Area of the Yellow River on the Qinghai–Tibet Plateau, China, and their eco-environmental impacts[J]. Environmental Research Letters, 2009, 4(4): 045206. doi: 10.1088/1748-9326/4/4/045206 [3] IMMERZEEL W W, van BEEK L P H, BIERKENS M F P. Climate change will affect the Asian water towers[J]. Science, 2010, 328(5984): 1382-1385. doi: 10.1126/science.1183188 [4] ZHANG X Y, WANG Y Q, ZHANG X C, et al. Carbonaceous aerosol composition over various regions of China during 2006[J]. Journal of Geophysical Research: Atmospheres, 2008, 113(D14):doi:10.1029/2007JD009525. [5] KASPARI S D, SCHWIKOWSKI M, GYSEL M, et al. Recent increase in black carbon concentrations from a Mt. Everest ice core spanning 1860-2000 AD[J]. Geophysical Research Letters, 2011, 38(4),doi:10.1029/2007JD009525. [6] CHEN P F, KANG S C, BAI J K, et al. Yak dung combustion aerosols in the Tibetan Plateau: Chemical characteristics and influence on the local atmospheric environment[J]. Atmospheric Research, 2015, 156: 58-66. doi: 10.1016/j.atmosres.2015.01.001 [7] WANG R, TAO S, WANG W T, et al. Black carbon emissions in China from 1949 to 2050[J]. Environmental Science & Technology, 2012, 46(14): 7595-7603. [8] BOND T C, DOHERTY S J, FAHEY D W, et al. Bounding the role of black carbon in the climate system: A scientific assessment[J]. Journal of Geophysical Research:Atmospheres, 2013, 118(11): 5380-5552. doi: 10.1002/jgrd.50171 [9] BOND T C, ZARZYCKI C, FLANNER M G, et al. Quantifying immediate radiative forcing by black carbon and organic matter with the Specific Forcing Pulse[J]. Atmospheric Chemistry and Physics, 2011, 11(4): 1505-1525. doi: 10.5194/acp-11-1505-2011 [10] QU B, MING J, KANG S C, et al. The decreasing albedo of the Zhadang glacier on western Nyainqentanglha and the role of light-absorbing impurities[J]. Atmospheric Chemistry and Physics, 2014, 14(20): 11117-11128. doi: 10.5194/acp-14-11117-2014 [11] CUI X J, WANG X F, YANG L X, et al. Radiative absorption enhancement from coatings on black carbon aerosols[J]. Science of the Total Environment, 2016, 551/552: 51-56. doi: 10.1016/j.scitotenv.2016.02.026 [12] ZHANG Y L, GAO T G, KANG S C, et al. Albedo reduction as an important driver for glacier melting in Tibetan Plateau and its surrounding areas[J]. Earth-Science Reviews, 2021, 220: 103735. doi: 10.1016/j.earscirev.2021.103735 [13] 姜璐, 邢冉, 陈兴鹏, 等. 青藏高原农区农户的家庭能源消费研究[J]. 地理科学, 2020, 40(3): 447-454. JIANG L, XING R, CHEN X P, et al. Rural household energy consumption of farmers in the Qinghai-Tibet Plateau[J]. Scientia Geographica Sinica, 2020, 40(3): 447-454 (in Chinese).
[14] LIU H K, WANG Q Y, XING L, et al. Measurement report: Quantifying source contribution of fossil fuels and biomass-burning black carbon aerosol in the southeastern margin of the Tibetan Plateau[J]. Atmospheric Chemistry and Physics, 2021, 21(2): 973-987. doi: 10.5194/acp-21-973-2021 [15] YANG J H, KANG S C, JI Z M. Critical contribution of South Asian residential emissions to atmospheric black carbon over the Tibetan Plateau[J]. The Science of the Total Environment, 2020, 709: 135923. doi: 10.1016/j.scitotenv.2019.135923 [16] TIAN J E, CHOW J C, CAO J J, et al. A biomass combustion chamber: Design, evaluation, and a case study of wheat straw combustion emission tests[J]. Aerosol and Air Quality Research, 2015, 15(5): 2104-2114. doi: 10.4209/aaqr.2015.03.0167 [17] CAO J J, WANG Q Y, LI L, et al. Evaluation of the Oxidation Flow Reactor for particulate matter emission limit certification[J]. Atmospheric Environment, 2020, 224: 117086. doi: 10.1016/j.atmosenv.2019.117086 [18] LI L, HUANG Z X, DONG J G, et al. Real time bipolar time-of-flight mass spectrometer for analyzing single aerosol particles[J]. International Journal of Mass Spectrometry, 2011, 303(2/3): 118-124. [19] WANG H L, AN J L, SHEN L J, et al. Mixing state of ambient aerosols in Nanjing city by single particle mass spectrometry[J]. Atmospheric Environment, 2016, 132: 123-132. doi: 10.1016/j.atmosenv.2016.02.032 [20] SONG X H, HOPKE P K, FERGENSON D P, et al. Classification of single particles analyzed by ATOFMS using an artificial neural network, ART-2A[J]. Analytical Chemistry, 1999, 71(4): 860-865. doi: 10.1021/ac9809682 [21] ZHANG X H, XU J Z, ZHAI L X, et al. Characterization of aerosol properties from the burning emissions of typical residential fuels on the Tibetan Plateau[J]. Environmental Science & Technology, 2022, 56(20): 14296-14305. [22] HEALY R M, HELLEBUST S, KOURTCHEV I, et al. Source apportionment of PM2.5 in Cork Harbour, Ireland using a combination of single particle mass spectrometry and quantitative semi-continuous measurements[J]. Atmospheric Chemistry and Physics, 2010, 10(19): 9593-9613. doi: 10.5194/acp-10-9593-2010 [23] CHRISTENSEN K A, STENHOLM M, LIVBJERG H. The formation of submicron aerosol particles, HCl and SO2 in straw-fired boilers[J]. Journal of Aerosol Science, 1998, 29(4): 421-444. doi: 10.1016/S0021-8502(98)00013-5 [24] LIANG Z C, ZHOU L Y, INFANTE CUEVAS R A, et al. Sulfate formation in incense burning particles: A single-particle mass spectrometric study[J]. Environmental Science & Technology Letters, 2022, 9(9): 718-725. [25] HUO J T, LU X H, WANG X N, et al. Online single particle analysis of chemical composition and mixing state of crop straw burning particles: From laboratory study to field measurement[J]. Frontiers of Environmental Science & Engineering, 2016, 10(2): 244-252. [26] KANG E, LEE M, BRUNE W, et al. Photochemical aging of organic and inorganic ambient aerosol from the Potential Aerosol Mass (PAM) reactor experiment in East Asia[J]. Atmospheric Chemistry and Physics, 2017,DOI:10.5194/acp-2016-1133 [27] GUO X, WANG X Q, DAI W T, et al. Effects of atmospheric aging processes on carbonaceous species and water-soluble inorganic ions in biomass burning aerosols[J]. Atmospheric Environment, 2022, 288: 119322. doi: 10.1016/j.atmosenv.2022.119322 [28] SUN J X, SUN Y L, XIE C H, et al. The chemical composition and mixing state of BC-containing particles and the implications on light absorption enhancement[J]. Atmospheric Chemistry and Physics, 2022, 22(11): 7619-7630. doi: 10.5194/acp-22-7619-2022 [29] MOFFET R C, de FOY B, MOLINA L T, et al. Measurement of ambient aerosols in northern Mexico City by single particle mass spectrometry[J]. Atmospheric Chemistry and Physics, 2008, 8(16): 4499-4516. doi: 10.5194/acp-8-4499-2008 [30] LIN Q H, BI X H, ZHANG G H, et al. In-cloud formation of secondary species in iron-containing particles[J]. Atmospheric Chemistry and Physics, 2019, 19(2): 1195-1206. doi: 10.5194/acp-19-1195-2019 [31] 牟莹莹, 楼晟荣, 陈长虹, 等. 利用SPAMS研究上海秋季气溶胶污染过程中颗粒物的老化与混合状态[J]. 环境科学, 2013, 34(6): 2071-2080. MU Y Y, LOU S R, CHEN C H, et al. Aging and mixing state of particulate matter during aerosol pollution episode in autumn Shanghai using a single particle aerosol mass spectrometer (SPAMS)[J]. Environmental Science, 2013, 34(6): 2071-2080(in Chinese).
[32] LI L, WANG Q Y, ZHANG Y, et al. Impact of reduced anthropogenic emissions on chemical characteristics of urban aerosol by individual particle analysis[J]. Chemosphere, 2022, 303: 135013. doi: 10.1016/j.chemosphere.2022.135013 [33] QI X, ZHU S P, ZHU C Z, et al. Smog chamber study of the effects of NO x and NH3 on the formation of secondary organic aerosols and optical properties from photo-oxidation of toluene[J]. Science of the Total Environment, 2020, 727: 138632. doi: 10.1016/j.scitotenv.2020.138632 [34] 张颖, 孔少飞, 郑煌, 等. 牛粪燃烧实时排放挥发性有机物特征研究[J]. 中国环境科学, 2020, 40(5): 1932-1939. ZHANG Y, KONG S F, ZHENG H, et al. Real-time emission of volatile organic compounds from cow dung combustion[J]. China Environmental Science, 2020, 40(5): 1932-1939 (in Chinese).
[35] WANG G H, CHENG C L, MENG J J, et al. Field observation on secondary organic aerosols during Asian dust storm periods: Formation mechanism of oxalic acid and related compounds on dust surface[J]. Atmospheric Environment, 2015, 113: 169-176. doi: 10.1016/j.atmosenv.2015.05.013 [36] WANG G H, KAWAMURA K, CHENG C L, et al. Molecular distribution and stable carbon isotopic composition of dicarboxylic acids, ketocarboxylic acids, and α-dicarbonyls in size-resolved atmospheric particles from Xi'an City, China[J]. Environmental Science & Technology, 2012, 46(9): 4783-4791. [37] CHENG C L, LI M, CHAN C K, et al. Mixing state of oxalic acid containing particles in the rural area of Pearl River Delta, China: Implications for the formation mechanism of oxalic acid[J]. Atmospheric Chemistry and Physics, 2017, 17(15): 9519-9533. doi: 10.5194/acp-17-9519-2017 [38] LI M, BAO F X, ZHANG Y, et al. Photochemical aging of soot in the aqueous phase: Release of dissolved black carbon and the formation of 1O2[J]. Environmental Science & Technology, 2019, 53(21): 12311-12319. [39] LI M, BAO F X, ZHANG Y, et al. Role of elemental carbon in the photochemical aging of soot[J]. Proceedings of the National Academy of Sciences of the United States of America, 2018, 115(30): 7717-7722. [40] ZHANG G H, FU Y Z, PENG X C, et al. Black carbon involved photochemistry enhances the formation of sulfate in the ambient atmosphere: Evidence from in situ individual particle investigation[J]. Journal of Geophysical Research: Atmospheres, 2021, 126(19), doi. org/10.1029/2021JD035226. -






 下载:
下载: