-
中水是国际公认的第二水源,但目前中水处理后应用的一个主要问题还是氨氮和总磷含量过高,而这部分物质在太阳光的照射下会滋生大量的微生物,使水体发臭,在硝化细菌的作用下会转化成硝酸和亚硝酸,从而影响中水的回用[1-2]。目前脱氮除磷的方法主要有化学沉淀法,物理吸附法和生物法,虽然各有特点,但都不同程度存在设备昂贵、运行费用高、能耗高和易造成二次污染等问题[3]。
硫酸亚铁(FeSO4)絮凝所具有的水质净化效能已在杀菌消毒、氧化除污染、混凝助凝等方面通过大量的工程应用得到了证实。GEORGIOU et al[4]研究了使用FeSO4絮凝处理绵纺织废水的色度和COD,用石灰将废水pH调节至9.0左右,绵纺织废水的色度去除率达到了70%,COD去除率达到50%。ZHU et al [5]使用FeSO4·7H2O处理锑矿的选矿废水,研究了絮凝剂用量,pH值和反应时间对废水中锑的去除效率。得到当七水合硫酸亚铁的用量为0.075∶100,pH为7,絮凝时间为180 min时,锑的去除率达到99.86%。
相比于传统的水处理技术,电化学絮凝在外电场的作用下,金属阳极氧化生成金属阳离子,经过水解和聚合生成具有凝聚和吸附作用的氢氧化物,以此来去除水中的污染物,因为不引入其他化学物质,且处理效率高、操作简便、占地面积小,被用于处理各种废水[6-8]。BAYRAMOGLU et al [9]研究表明,相比于化学混凝,电絮凝处理纺织废水的COD和浊度的效率更高,消耗的材料和产生的污泥更少,同时产生的多核羟基化合物对水中的磷有较好的吸附效果。
文章通过小试考察不同FeSO4投加量、pH值、通电时间和电压分别研究了FeSO4絮凝和电化学絮凝对中水中低浓度的氨氮和总磷的去除效果,以及电化学为主体结合FeSO4絮凝优化对中水脱氮除磷的效果,为中水的深度净化提供基础数据以指导工程实践。
全文HTML
-
实验所用的试剂与仪器,见表1。
-
实验用水样为西安工程大学污水处理系统的中水,氨氮含量为0.8 mg/L,总磷含量为0.4 mg/L,pH为7。所排出中水水质符合城市污水厂处理排放标准一级A标准。
-
取实验水样200 mL,投加一定量的FeSO4,使用六联电动搅拌器(100 r/min)搅拌5 min后,静置沉降1 h使用滤纸过滤絮体,再滴加NaOH溶液去除水中多余的铁离子,取上清液测定氨氮及总磷。
-
取中水300 mL于烧杯中,测定铝电极在一定电压和不同通电时间下对原水电解絮凝一定时间后水中氨氮和总磷的浓度。电解槽容积为500 mL,板间距1 cm,极板有效面积为10 cm2,絮凝后将水样过滤测定上清液中氨氮和总磷的浓度。
-
首先使用FeSO4对中水进行预处理,取上清液过滤后倒入500 mL烧杯中,以铝电极进行电化学絮凝,极板间距为10 mm,极板有效面积为10 cm2。电解过程结束后,取出极板去除上层絮凝悬浮物,静置一段时间后过滤,向滤液加入一定量的氢氧化钠溶液去除多余的铁离子,静置20 min取上清液进行测量。
-
水样中氨氮的浓度采用氨氮试剂盒测定法,总磷的浓度采用总磷测定试剂盒检测法。
1.1. 试剂与仪器
1.2. 实验用水
1.3. 实验方法
1.3.1. 硫酸亚铁絮凝实验
1.3.2. 电化学絮凝实验
1.3.3. FeSO4/电化学絮凝体系处理中水
1.4. 分析方法
-
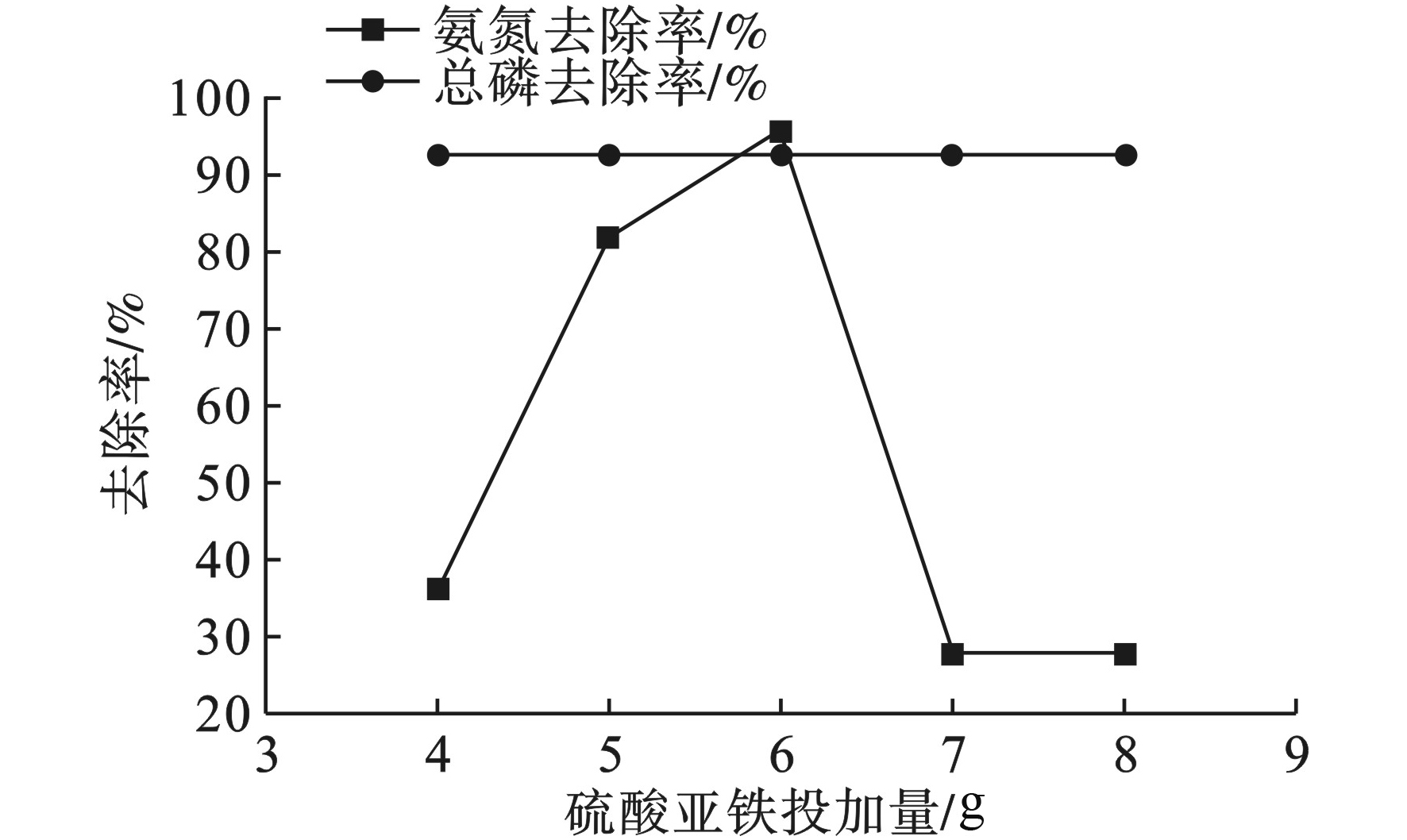
FeSO4的投加量对氨氮和总磷去除率的影响,见图1。
图1可见,氨氮去除率随硫酸亚铁投加量的增加先升高后降低。在pH为7,投加5 mL质量分数为6 %(56.6 g/L)的FeSO4,搅拌5 min后,再静置沉降1 h,氨氮去除率最大可达95.45 %。因为Fe2+氧化生成的Fe3+具有空轨道,而水中游离的NH3和NH4+中的氮有一个孤对电子对,因此会被Fe3+所吸附,最终被生成的多核羟基络合物絮凝沉淀。当FeSO4反应系统接近饱和时,过量的Fe2+反而会氧化已生成的Fe3+多核羟基络合物,影响氨氮在水体中被絮凝沉降的效果[10],所以实验结果成正态分布。
化学除磷是通过混凝剂中Fe2+与水中溶解氧发生氧化反应生成可以形成多核羟基络合物的Fe3+,同时Fe2+和Fe3+都可以与PO43−生成难溶性磷酸盐,见式(1)、(2),并利用络合物表面的吸附作用去除水中的磷。而由于原水中磷元素的含量是微量的,所以水样中的总磷较容易被去除,去除率为92.5%不变,因此确定FeSO4投加量为6%。
-
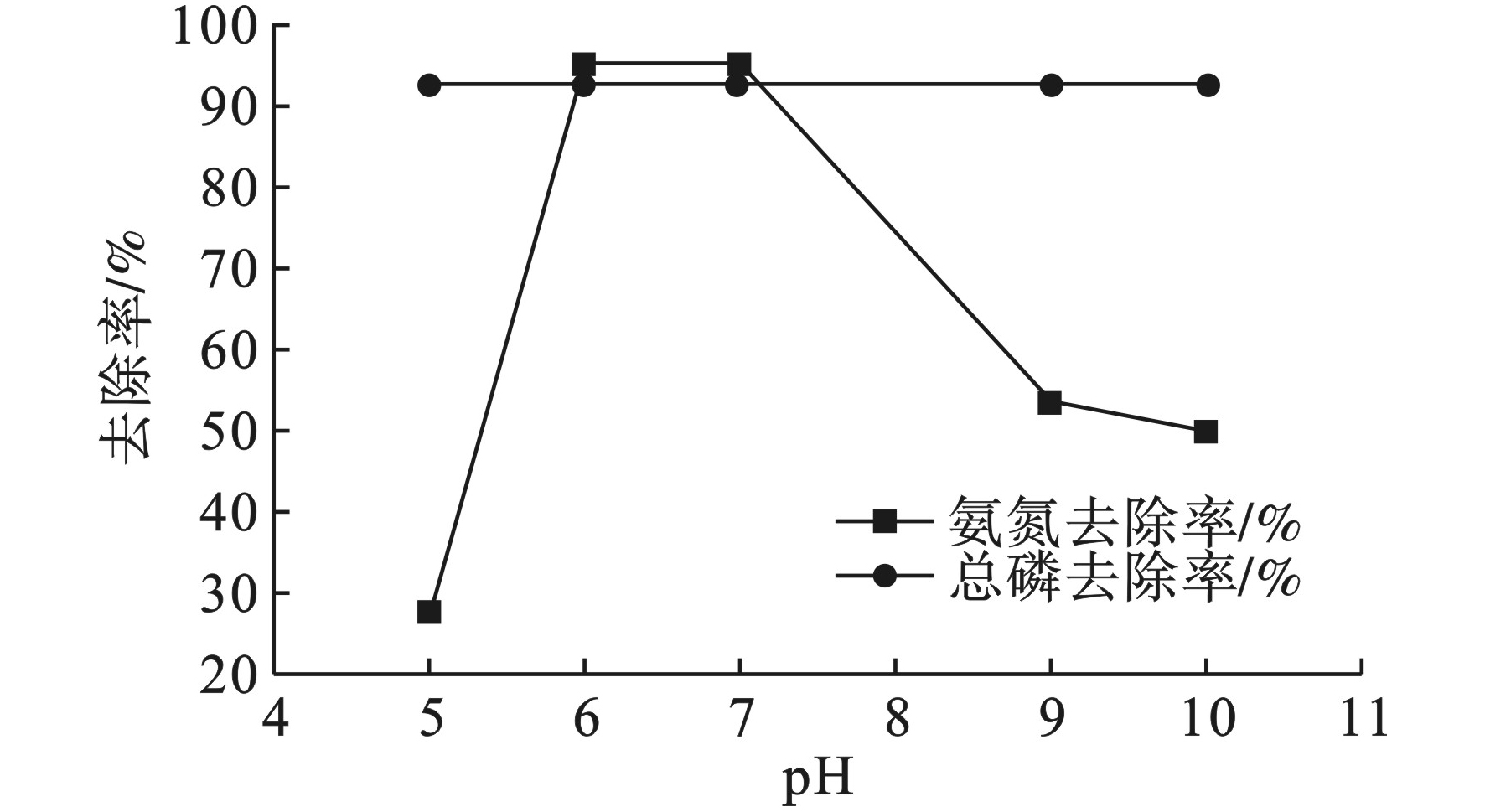
氨氮和总磷去除率随pH值变化趋势,见图2。
图2可见,当FeSO4投加量的质量分数为6%时,氨氮去除率在pH值6~7时达到最大为95.45%,总磷去除率为92.5%。pH值是一个比较关键的影响因素,游离NH3和NH4+见式(3)在水中保持平衡关系,当pH较高时水中NH3较多,当pH较低时,水中NH4+较多[11]。但过高和过低的pH会影响FeSO4形成多核羟基络合物的效果[12],从而影响氨氮的去除效果。所以最佳pH为6~7。因为中水中磷的浓度较低且去除方式为直接化学沉淀,所以pH对磷的去除影响不大。
-
不同通电时间和电压对水中污染物去除率的影响,见图3。
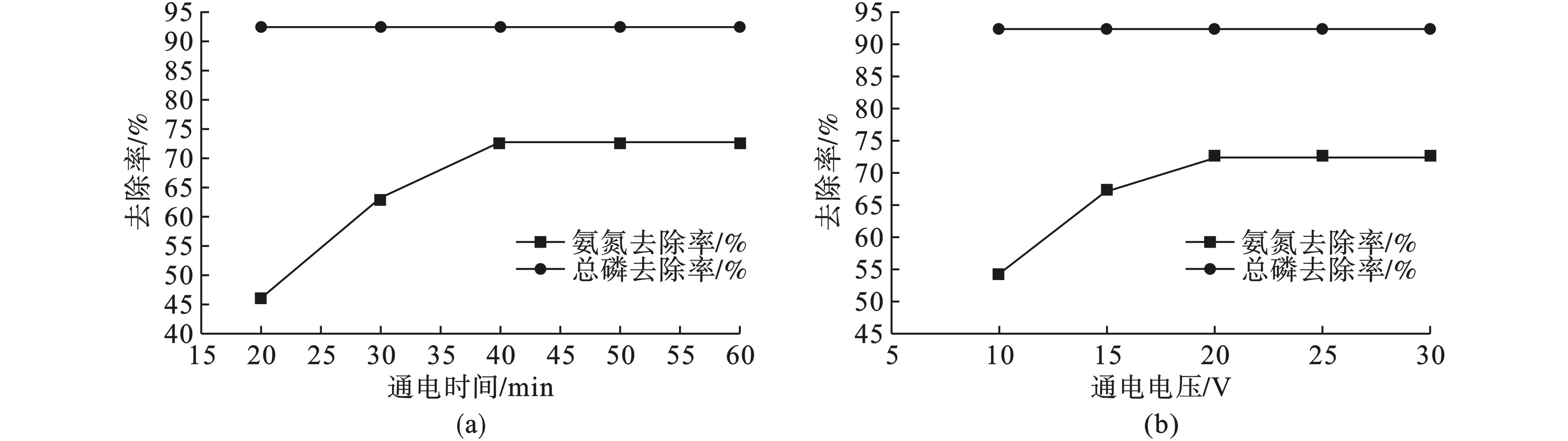
在20 V通电电压下,水中氨氮和总磷去除率随通电时间的变化情况,见图3(a)。
图3(a)可见,氨氮去除率随通电时间的增加而增加。当通电电压为20 V,通电时间为40 min时,氨氮去除率可达72.7%,总磷去除率为92.5%,随后再增加通电时间氨氮去除率基本不变。因为铵离子通过电极的析氧反应所生成的羟基自由基的氧化作用生成N2而得以去除,但中性和酸性条件下的分解率远低于碱性条件下的分解率[13-14],所以对氨氮的去除率相比硫酸亚铁絮凝较低。而过长的电解时间,会产生过量的Al3+,生成大量不溶物质所包裹的胶粒,导致阳极钝化,增加电极的电阻,影响极板间的电流密度,从而导致了电化学反应发生缓慢、絮凝性能不再增加,所以在通电一段时间后会出现氨氮去除率趋于平衡。
水中的磷以无机态的形式存在为主,当采用电絮凝技术除磷时,铝作为阳极材料在电解时生成的金属阳离子或其水合物与水中的磷酸盐形成沉淀而去除水中的磷[15]。反应原理见式(4)、(5)。
因此,确定40 min为最佳反应时间。
-
在通电40 min,水中氨氮和总磷去除率随通电电压变化所产生的影响,见图3(b)。
图3(b)可见,氨氮去除率随通电电压的增加而增大,在通电电压为20 V,通电时间40 min时,氨氮的去除率为72.7%,总磷去除率为92.5%。随后再增大电压氨氮去除率保持不变。因为电压会影响电解过程中羟基铝化物的产生量、气泡的产生速率以及羟基铝化物与氨氮和磷的混合速率,当电压增加到一定值后以上指标将不再变化[16]。因此电压应选择20 V,通电时间40 min。
-
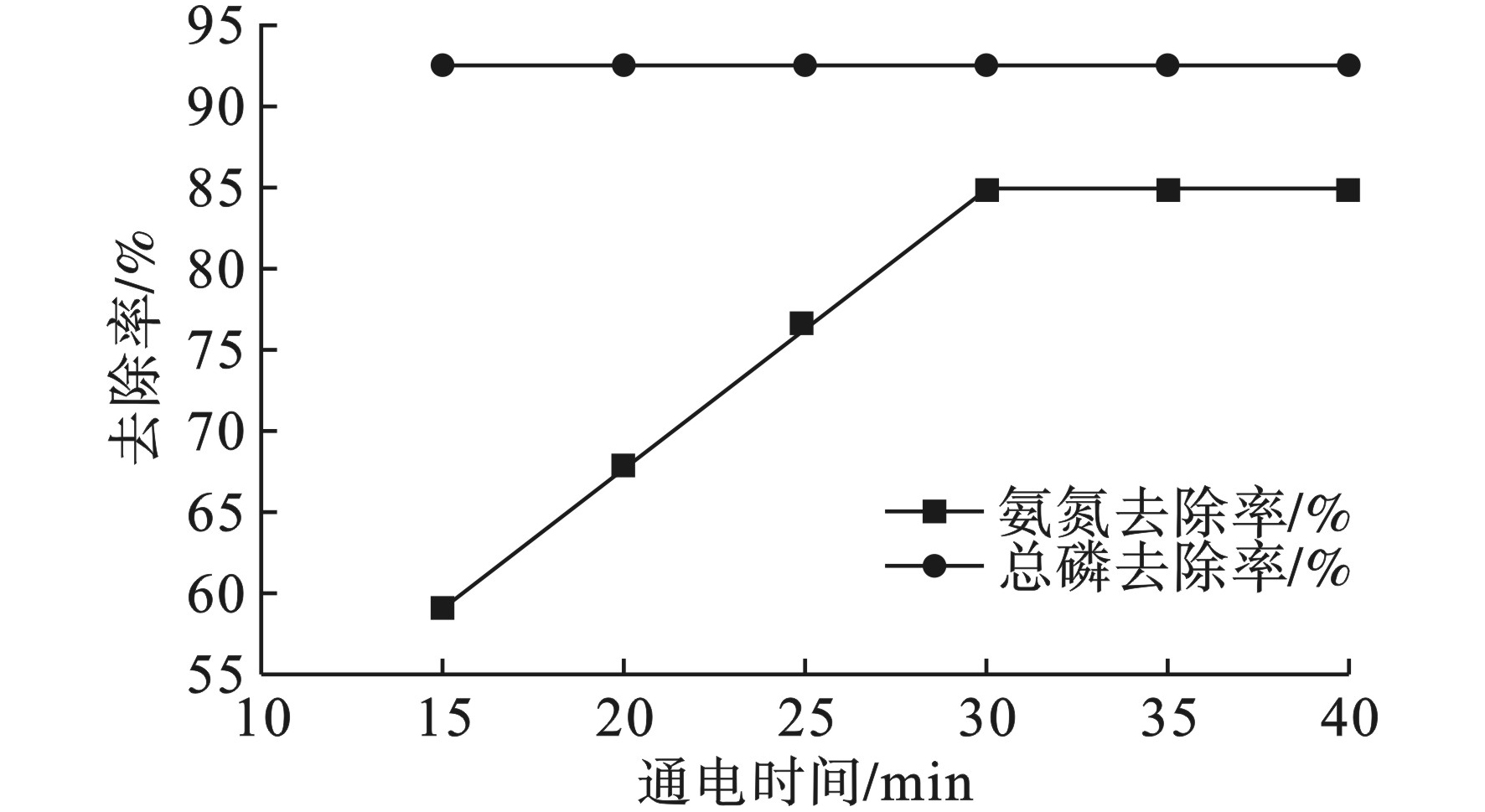
使用FeSO4絮凝法在pH为6~7,处理后再使用电絮凝进行深度处理,处理效果,见图4。
图4可见,在FeSO4投加量为6%,通电电压20 V时,氨氮的去除率随通电时间的增加而增大。氨氮去除率在30 min时趋于稳定,达到84.9%,总磷去除率为92.5%。
FeSO4/电絮凝体系对氨氮的去除率高于单纯使用电絮凝的72.7%,但小于单纯使用FeSO4絮凝法时氨氮95.45%的去除率。原因可能是FeSO4在水中电离后产生的羟基铁离子与阳极产生的羟基铝化物发生了互相湮灭的反应,即互为絮凝,使得氨氮的去除率相比于单纯使用FeSO4的去除率有所下降。
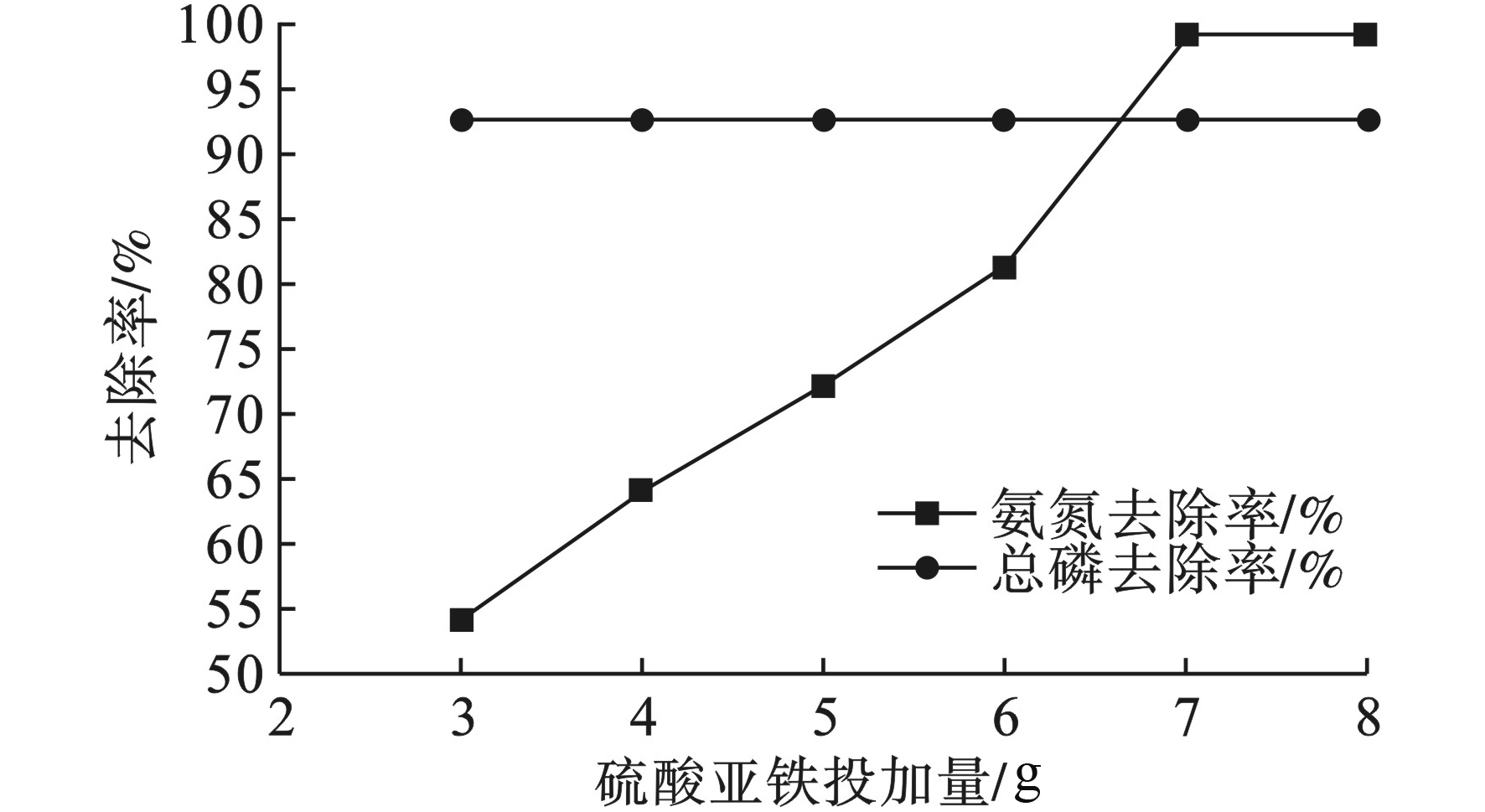
在改变FeSO4投加量后依然保持通电时间30 min,通电电压20 V。FeSO4投加量对FeSO4/电絮凝体系处理效果的影响,见图5。
图5可见,氨氮去除率随FeSO4投加量增加而增大。当FeSO4的投加量为7%时,氨氮去除率达到99.09%,总磷去除率为92.5%。随后再增加FeSO4的投加量,氨氮和总磷去除率保持不变。氨氮去除率得到提升的原因可能是加大FeSO4投加量后,水中过量的Fe2+与阳极电离水所产生的O2,H2O2发生了类Fenton反应,羟基自由基的强氧化性使得中水中低浓度氨氮的去除率有了较大的提高,同时也去除了水中低浓度的磷[16-17]。
以上结果表明:Fe3+具有的空轨道可以吸附游离NH3和NH4+中氮的孤对电子对,而Fe2+和Fe3+都可以与PO43-生成难溶性磷酸盐,最终被生成的多核羟基络合物絮凝沉淀,从而去除中水中低浓度的氨氮和磷。
电化学絮凝处理水中低浓度氨氮是因为铵离子通过电极的析氧反应所生成的羟基自由基的氧化作用生成氮气而得以去除,去除水中低浓度的磷是通过铝作为阳极材料在电解时生成的金属阳离子或其水合物与水中的磷酸盐形成沉淀。
而FeSO4/电絮凝体系对水中氨氮处理效果的提升可能是因为水中过量的Fe2+与阳极电离水所产生的O2,H2O2发生了类Fenton反应,产生了更多的羟基自由基氧化了水中的氨氮。
2.1. 硫酸亚铁絮凝法
2.1.1. FeSO4投加量对中水处理效果的影响
2.1.2. pH值对FeSO4处理中水的影响
2.2. 电化学絮凝法
2.2.1. 不同通电时间对去除率的影响
2.2.2. 不同电压对去除率的影响
2.3. 硫酸亚铁结合电化学水处理法
-
1)在pH为6~7的200 mL水样中,投加5 mL浓度为6%的FeSO4搅拌5 min,再静置沉降1 h,氨氮去除率达到95.45%,总磷去除率达到92.5%。FeSO4的投加量和原水pH都会影响水中低浓度氨氮和总磷的去除率,其关系基本都符合正态分布。
2)中水中氨氮的去除率随着电絮凝的通电时间和通电电压的增加而增加,在通电电压为20 V,通电时间40 min时,氨氮去除率达到72.7%,最大且不再增加,总磷去除率达到92.5%。过长的电解时间和过大的电压都会使得阳极出现钝化现象,增加电极的电阻,影响极板间的电流密度,从而导致了絮凝性能不再增加、电化学反应发生缓慢。
3) FeSO4/电絮凝体系去除中水中低浓度的氨氮与总磷的效果有一定提升。在FeSO4投加量7 %,通电电压20 V,通电时间30 min时,氨氮去除率达到99.09%,总磷去除率达到92.5%。其原因可能是因为水中过量的Fe2+与阳极电离水所产生的O2,H2O2发生了类Fenton反应,产生了更多的羟基自由基氧化了水中的氨氮。



 下载:
下载: