UV/H2O2工艺降解环丙沙星的研究
Photodegradation of ciprofloxacin by UV/2O2 process
-
摘要: 采用UV/2O2工艺去除水体中的喹诺酮类抗生素环丙沙星(CIP)。考察了溶液pH值、2O2投加量以及水体基质对环丙沙星降解效率的影响,分析了降解产物的生成情况。研究表明,环丙沙星的降解符合拟一级反应动力学模型。降解速率受溶液pH值的影响,酸性及中性条件,有利于环丙沙星的降解。2O2投加量的增大,使得降解速率逐渐增大,但速率增幅逐渐变缓;最佳2O2/环丙沙星摩尔比为2 000。实际水体中存在的NOM、NO-3,促进了单独UV作用下,环丙沙星的降解。水体中的?OH焠灭剂,抑制了UV/2O2联合作用下,环丙沙星的降解;实际水体中的光解速率常数低于超纯水中的光解速率常数。GC-MS分析表明,UV/2O2工艺,使环丙沙星氧化降解生成氨基乙酸、丙二酸、丙三醇和对苯二甲酸等小分子有机物。Abstract: The photodegradation of ciprofloxacin in UV/2O2 process was investigated with a low-pressure UV lamp system. The influence of initial pH values, 2O2 concentration and water quality was discussed; proposed photoproducts of ciprofloxacin were also analyzed. Results show that ciprofloxacin displays a pH-dependent degradation and the neutral condition is more favorable. Reaction rates increased with the increasing of 2O2 concentration, and the optimal 2O2/ciprofloxacin molar ratio at which the highest reaction rate constant is reached is 2 000. In UV direct photolysis, photodegradation rates of ciprofloxacin in natural water are higher than in ultrapure water; while in UV/2O2 process, the photodegradation rates in natural water are lower than those in ultrapure water. Moreover, several photoproducts such as propanedioic acid, glycerin, 1, 4-benzenedicarboxylic acid, and some aliphatic acids etc. forming in the photodegradation of ciprofloxacin in UV/2O2 process are identified by GC-MS.
-
Key words:
- ciprofloxacin /
- UV/2O2 /
- kinetics /
- photodegradation
-
近20年来,我国在城镇水环境卫生方面取得了长足发展。截至2019年底,用于污水处理及其再生利用的市政公用设施建设投资已接近每年1 000亿元,较20年前翻了近10倍;全国城镇污水处理厂总数也由2000年初的481座增加至4 140座,并仍以每年约5%的增速增长[1]。然而,国内污水处理体量的迅猛提升并不代表污水处理技术水平的快速提高。随着国家/地方污水排放标准日益收紧,国内老旧污水处理厂普遍遇到稳定达标难、能耗药耗高等运营技术问题[2-3]。
基于活性污泥模型(activated sludge model,ASM)的生物建模技术经过30余年的发展,在国内已逐步应用于工程领域[4]。在污水厂运营优化方面,郝晓地等[5]利用代尔夫特-活性污泥联合代谢模型(TUD-ASM Model)模拟对比了UCT(University of Cape Town)工艺的优势,结果表明,在国内污水厂升级改造时通过强化反硝化除磷菌(Denitrifying Phosphorus removal Bacteria)的作用可实现碳源与除磷药剂的节省。郝二成等[6]利用GPS-X软件成功构建了大连某污水处理厂UCT工艺数学模型,模拟结果可为该厂运行优化提供曝气与加药控制方案,以实现节能降耗。然而,国内的模型研究仍以数据分析和理论探索为主,缺少应用型案例及对相关模拟结论的试验与验证,导致生物模型用于解决我国污水处理厂实际运营技术问题的经验不足,故国内亦尚未形成成熟的生物建模技术应用标准。
本研究以泗县污水处理厂改良型氧化沟工艺为案例,利用经过实践检验的系统性三步数据清洗方法完成了全厂生物建模与模型校正,结合模型校正参数实现了对污水处理厂的问题诊断,最后利用校正后的模型完成技术优化方案比选。通过实时跟踪改造效果,完成了模拟优化方案验证,以期为生物建模技术在国内污水处理厂问题诊断与运行优化应用中提供参考。
1. 材料和方法
1.1 污水处理厂概况
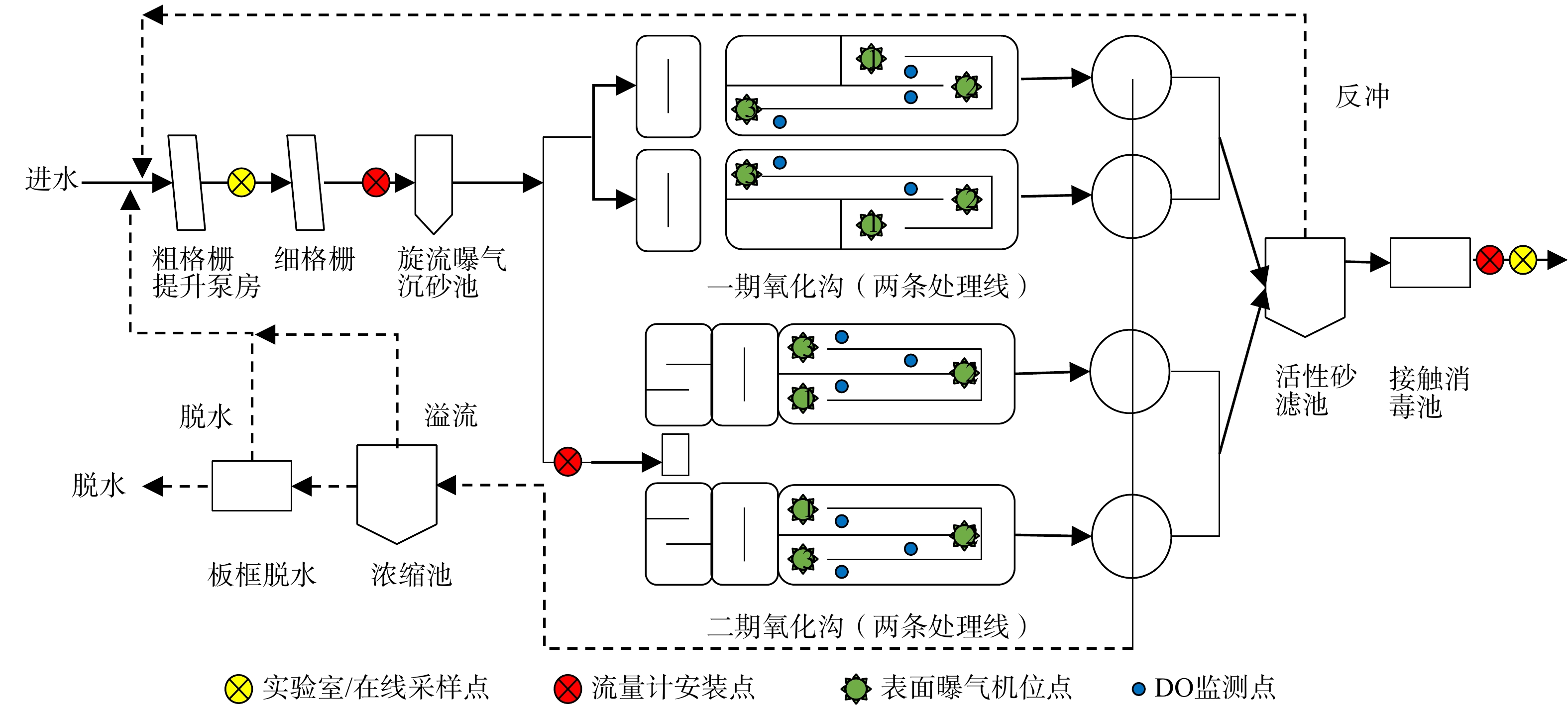
案例水厂为泗县污水处理厂,总设计规模40 000 m3·a−1,采用“改良型氧化沟+活性砂滤池”工艺,分2期共4条水处理线。其中,一期正式投产于2008年且原为Carrousel氧化沟工艺,2017年完成提标改造后,将氧化沟前端廊道分隔为独立缺氧池,并新建厌氧池,出水标准由一级B提升为一级A;同年,二期正式通水且主体工艺与提标后的一期基本一致。一、二期共用1套预处理单元,包括粗格栅、细格栅和旋流曝气沉砂池。污水经预处理后由管道重力自流至一、二期改良型氧化沟, 4组二沉池出水混合后经活性砂滤池统一排出。一、二期共用1条污泥处理线,剩余污泥均排入2座贮泥池静置2~4 h后,经污泥调理池投加脱水剂(CaO与FeCl3)进入板框压滤机排出,全厂工艺流程图详见图1。2017年至今,水厂总出水执行《城镇污水处理厂污染物排放标准》(GB 18918-2002)A标准,脱水污泥含水率<60%。
在水质水量方面,案例厂全部为生活污水且无工业废水掺杂,通过整理该厂2019年水质检测数据发现,该厂全年水质稳定、无明显波动,仅雨季期间(8月)平均进水水质偏低。这与旧城区采用雨污合流管网有关(见表1)。研究期间,水厂平均日处理水量为37 757 m3·d−1,约占设计能力的94.3%。此外,城区主要进水泵站均由水厂运行人员控制,因此,全年水量控制稳定,无明显冲击。
表 1 实验室2019年历史进水化验数据Table 1. Laboratory data of historical influent in 2019季度 月份 COD/(mg·L−1) 氨氮/(mg·L−1) 总氮/(mg·L−1) 总磷/(mg·L−1) 一季度 1 152 22.4 30.2 3.40 2 166 25.3 32.9 3.56 3 189 26.7 32.9 2.99 二季度 4 176 24.4 29.9 2.71 5 170 32.6 38.2 3.90 6 206 27.7 32.0 3.66 三季度 7 194 26.8 32.9 4.01 8 124 19.4 26.7 2.45 9 157 30.8 38.5 3.32 四季度 10 143 30.9 39.6 2.88 11 168 32.2 39.4 2.96 12 156 26.6 34.7 2.96 1.2 模型构建与数据清洗
本次采用的生物建模软件为EnviroSim Biowin 5.3,计算模型为其内嵌的活性污泥-厌氧消化联合代谢模型(Biowin-ASDM)。该模型以活性污泥模型(ASM)为核心并已广泛应用于国内外污水处理厂生物建模研究[7-10]。建模步骤采用经过实践检验的基于数据清洗的本土化生物建模[11]方法。本研究具体步骤包括如下3步。
1) 明确建模目标。结合案例厂提出的实际运行问题,如污泥浓度变化大、污泥脱水剂(CaO、FeCl3)药耗量高,确定本研究的建模目标为利用生物模型完成水厂问题诊断,确定最佳污泥浓度控制策略,提出综合调控手段,以解决脱水剂药耗过高问题,实现节能降耗。
2) 数据采集。建模数据源包括2020年1月至7月水质检测数据、SCADA系统在线流量数据及2020年7月16日至8月16日间3次补充采样化验数据。
3) 数据清洗。根据系统性3步清洗方法剔除大误差、闭合物料平衡、修正污泥组分,修正后的各平衡流量数据与污泥组分结果列于表2及表3。
表 2 物料平衡校核结果Table 2. Mass balance calculation results物料项目 原始值/(m3·d−1) RSD 修正值/(m3·d−1) 修正系数 修正方法 混合进水 37 757 8.21% 38 829 +2.83% 平衡值 总进水 − − 38 200 − 计算值 总出水 35 325 6.92% 38 189 +8.11% 计算值 进水(二期) 15 353 7.45% 17 000 +10.73% 平衡值 脱水泥量 13.7 40.46% 14 +2.19% 平衡值 剩余污泥量(一期) 131.5 105.83% 243 +84.79% 平衡值 剩余污泥量(二期) 139.2 123.04% 167 +19.97% 平衡值 污泥回流量(一期) − − 31757 − 计算值 污泥回流量(二期) 25 468 11.58% 26 833 +5.36% 平衡值 内回流量(一期) − − 52 000 − 计算值 内回流量(二期) − − 25 000 − 计算值 氧化沟内循环量(一期) − − 450 000 − 计算值 氧化沟内循环量(二期) − − 360 000 − 计算值 进脱水机泥量 − − 128 − 计算值 反冲洗水量 − − 231 − 计算值 脱水机脱水液 − − 115.9 − 计算值 除磷剂 1.44 14.05% 1.44 0% 测量值 碳源药剂 0.48 62.37% 0.48 0% 测量值 表 3 污泥组分修正结果Table 3. Correction results of sludge composition指标名称 样品名称 测量值/(g·g−1) 修正系数 修正值 国外参考标准范围[15] VSS/TSS 曝气池(一期) 0.284 +15.14% 0.327 0.71~0.82 VSS/TSS 剩余污泥(一期) 0.366 −10.66% 0.327 0.71~0.82 VSS/TSS 曝气池(二期) 0.280 +12.14% 0.314 0.71~0.82 VSS/TSS 剩余污泥(二期) 0.330 −4.84% 0.314 0.71~0.82 CODX/VSS 曝气池(一期) 2.08 −13.40% 1.80 1.32~1.42 CODX/VSS 剩余污泥(一期) 1.29 +39.53% 1.80 1.32~1.42 CODX/VSS 曝气池(二期) 2.24 −18.30% 1.83 1.32~1.42 CODX/VSS 剩余污泥(二期) 1.61 −13.66% 1.83 1.32~1.42 TKNX/CODX 曝气池(一期) 0.060 +3.38% 0.062 0.05~ 0.08 TKNX/CODX 剩余污泥(一期) 0.062 −0.76% 0.062 0.05~ 0.08 TKNX/CODX 曝气池(二期) 0.060 +4.04% 0.062 0.05~ 0.08 TKNX/CODX 剩余污泥(二期) 0.062 −0.579 0.062 0.05~ 0.08 TPX/CODX 曝气池(一期) 0.0333 +15.3% 0.038 4 0.035~0.055 TPX/CODX 剩余污泥(一期) 0.0380 +1.05% 0.038 4 0.035~0.055 TPX/CODX 曝气池(二期) 0.0308 +27.60% 0.039 3 0.035~0.055 TPX/CODX 剩余污泥(二期) 0.0360 +9.27% 0.039 3 0.035~0.055 表2所示的物料平衡校核结果表明,除剩余污泥流量外,各流量计读数可靠性较高。调查发现,该厂一、二期剩余污泥泵房的管式电磁流量计常年无维护记录,并且因二沉池底流污泥浓度波动大、日流量小且为非连续流,易导致流量计数据不准,该误差为污水处理厂常见数据误差来源之一[12]。表3中的污泥组分修正结果显示,该厂活性污泥VSS/TSS偏低,该现象在国内研究中普遍存在,其原因与除磷药剂过量投加及高进水无机颗粒ISS浓度有关[11,13]; 而CODX/VSS修正值高于国外参考值(此处“X”表示国际水协ASM模型中的通用符号,表示颗粒态物质,后同),表明单位质量污泥具有较高的有机质含量,进水中含有较多的高分子油脂类有机物(COD/VSS>2)可能是产生该现象的主要原因[14]。
2. 结果与讨论
2.1 模型校正与问题诊断
生物模型基于清洗后的数据并参考MEIJER提出的方法进行模型校正[15],再将2020年1—7月水质检测数据及主要操作记录输入校正模型,完成模型验证。除2个基本水质特征参数外,该模型仅校正5个计量学参数和4个动力学参数(见表4),即可实现主要污染物去除率模拟误差≤4.5%(见表5),说明基于清洗数据校正后的模型可以准确拟合实际污水处理厂的污泥性质与处理效果,模型可靠性高[16]。表4校正参数均根据表3实测污泥组分及物料平衡校核结果进行调整,相关参数均在正常调整范围。需要说明的是,FUP与FUS均高于缺省值表明进水COD中的惰性组分含量高,导致系统无效的活性污泥浓度增加,为保证有效活性污泥含量,该厂实际氧化沟MLSS应高于设计值;在动力学参数中,
KHO2 表 4 校正参数表Table 4. Calibration parameter table校正参数名称 参数类型 参数描述 校正原因 校正值 缺省值 单位 FUP 水质特征 惰性颗粒COD 拟合污泥产量 0.18 0.13 g·g−1 FUS 水质特征 惰性可溶COD 拟合出水COD 0.09 0.05 g·g−1 INOHO 计量学 OHO中N 拟合污泥N组分 0.06 0.07 mg·mg−1 INEND 计量学 内源产物中N 拟合污泥N组分 0.06 0.07 mg·mg−1 XI∶VSS 计量学 惰性颗粒COD与VSS质量之比 拟合污泥TSS平衡 2.35 1.6 g·g−1 XS∶VSS 计量学 可降解颗粒COD与VSS质量之比 拟合污泥TSS平衡 1.05 1.6 g·g−1 KPFe 计量学 化学沉淀物中[Fe]∶[P] 拟合出水PO4浓度 2.10 1.6 mol·mol−1 μA 动力学 AOB最大比增长速率 拟合出水NH3浓度 0.95 0.9 mg·L−1 KHO2 动力学 OHO对DO半饱和系数 拟合TN平衡 0.30 0.15 mg·L−1 KNO2 动力学 NOB对DO的半饱和系数 拟合出水NO2浓度 0.25 0.5 mg·L−1 αF 动力学 氧气传质效率 拟合DO梯度 0.65 0.5 − 表 5 污染物去除率的实际值与模拟结果对比Table 5. Comparison of simulation results数据来源 指标 实际值 模拟值 误差 一期 TSS 93.9% 97.2% 3.3% TCOD 87.3% 88.6% 1.3% TN 77.5% 76.1% −1.4% TP 91.6% 92.0% 0.4% 二期 TSS 89.8% 92.3% 2.5% TCOD 81.9% 86.4% 4.5% TN 68.6% 68.9% 0.3% TP 81.7% 82.9% 1.2% 全厂 TSS 93.6% 96.5% 2.9% TCOD 87.3% 88.4% 1.1% TN 74.1% 73.1% −1.0% TP 89.3% 90.2% 0.9% 生物模型确定后,污水处理厂的实际运行状况及边界条件亦随之确定[17]。与根据经验或传统计算方法分析水厂运行问题的方式不同,校正后的生物模型可以对整条工艺线中各点的水质、水量、工况实现量化评估。因此,利用校正与验证后的生物模型计算核心工艺运行参数,结果列于表6。通过对比表6中污泥负荷、容积负荷、反应区容积比、沟内流速4组工艺参数可发现,尽管该厂一、二期工艺的改造/设计路线为AAO工艺(在氧化沟式曝气池基础上,增加独立厌氧池与缺氧池,并增加硝化液回流),但该厂实际运行控制方式仍接近氧化沟工艺的设计参数[18-19]。结合表4模型校正对
KHO2 KHO2 表 6 生物模型工艺参数分析Table 6. Analysis of process parameters of the biological model参数名称 单位 一期 二期 氧化沟设计参考值 AAO设计参考值 污泥负荷(以BOD5计) kg·(kg·d)−1 0.032 0.027 0.03~0.08 0.1~0.2 容积负荷(以BOD5计) kg·(m3·d)−1 0.16 0.14 0.2~0.4 1.0~1.8 污泥质量浓度 mg·L−1 4 843 5 265 2 500~4 500 2 500~4 500 反应区容积比 − 1∶2∶8 1∶2∶6 − 1∶1∶5 生化池总容积 m3 12550 13320 污泥龄 d 18.4 28.5 >15 10~20 处理水量分配 % 55.5 44.5 污泥回流比 % 125 160 50~100 内回流比 % 230~280 140~200 200 沟内流速 m·s−1 0.21 0.16 0.25 表曝机功率 kW 40 45 此外,通过对比污泥龄、水量分配、污泥回流比、内回流比、表曝机功率5组工艺参数可发现如下结论。1) 2期工艺污泥龄不同。一方面,由于水量分配不均造成一期污泥产量高于二期;另一方面,在相同的排泥策略下,为控制相同/接近的污泥浓度,一期排泥量显著高于二期(表2)。一、二期实际污泥龄相差近10 d。2)污泥回流比偏高。一、二期污泥回流比均长期>120%且二期高于一期。3)内回流比不同。由于内回流硝化液在沟内流速差的作用下自流至缺氧区,因此两期工艺内回流比理论上无法控制。4)曝气效率不同。一二期表曝机铭牌功率均为45 kW,但拟合结果表明一期曝气功率仅为二期90%,在水量分配不均情况下可能导致脱氮效率的显著差异。因此,应进一步利用模型设计情景模拟分析,对上述问题进行量化评估。
2.2 情景分析与模拟优化
2.2.1 泥龄影响的量化分析
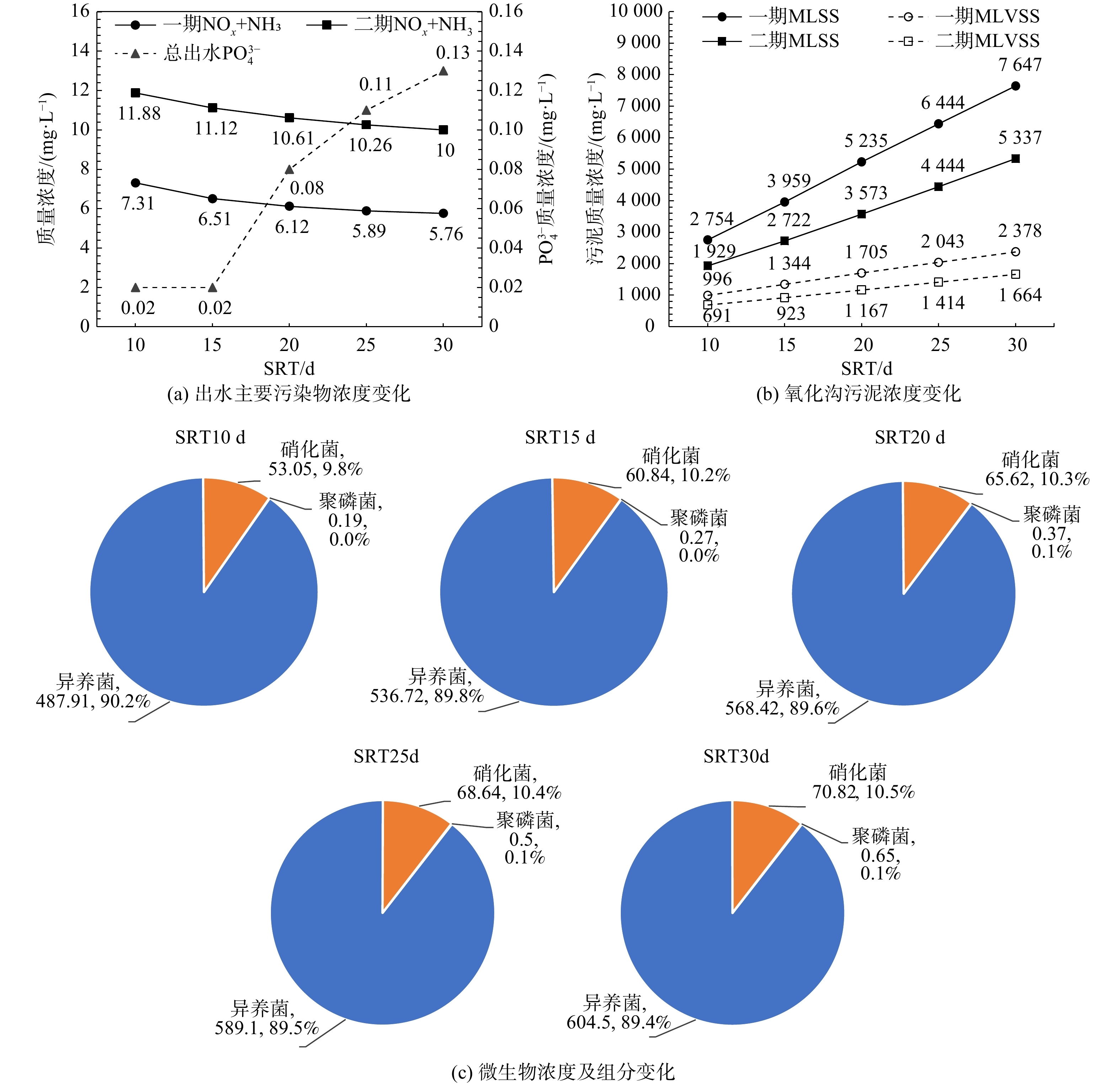
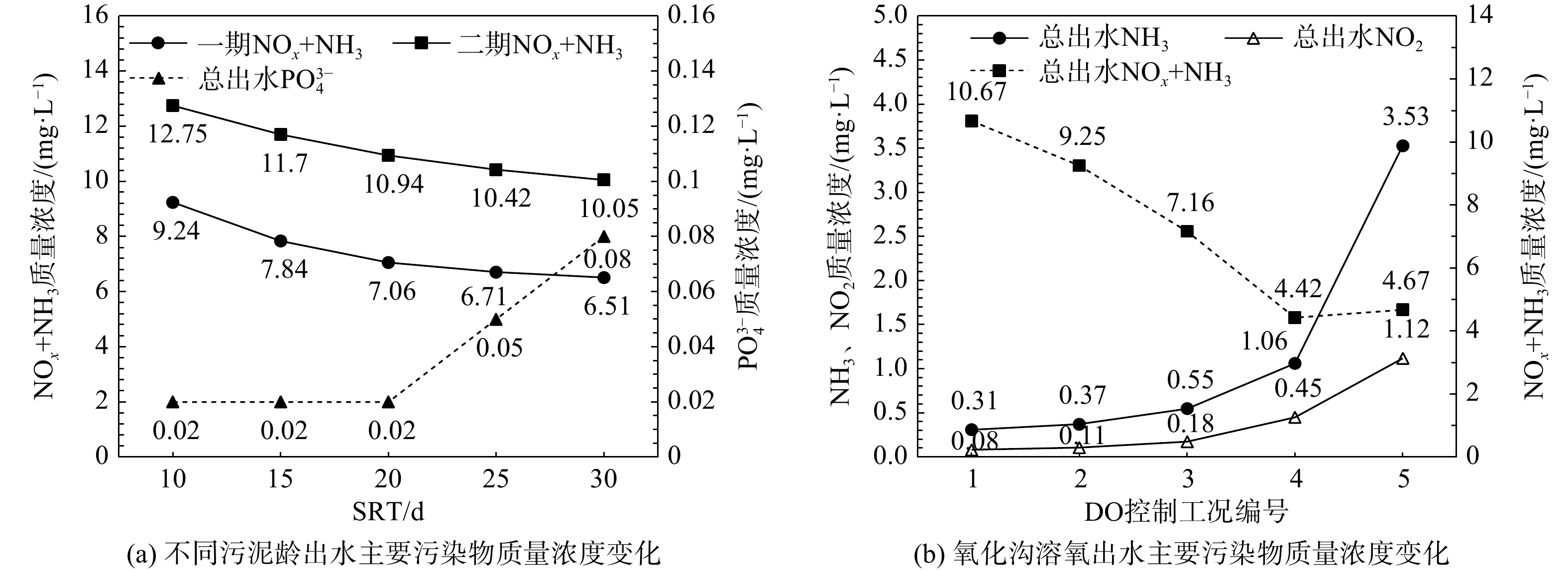
合理的污泥龄(sludge retention time,SRT)控制是活性污泥系统最基本和最重要的策略,因此,首先利用验证后的模型分别控制SRT为10、15、20、25、30 d。在平均进水水质及正常运行工况下,稳态模拟一、二期出水主要污染物浓度变化及污泥浓度变化,结果见图2(a)和图2(b)。同时,将不同SRT时异养菌(XOHO)、硝化菌(XAUT)、聚磷菌(XPAO)浓度及组分变化绘制于图2(c)。图2(a)和图2(b)表明,随着SRT的升高,污泥浓度逐渐升高,出水溶解性氮(氨氮+硝酸氮)浓度逐渐降低,出水磷酸盐浓度在SRT≤15 d时浓度不变,在SRT>15 d时浓度迅速升高。结合图2(c)可发现,随着SRT得升高,活性污泥中微生物浓度(OHO、AUT、PAOs)均明显提高,但微生物组分并未发生显著变化。这是由于模拟最小泥龄10 d已大于当前工况的硝化最小泥龄,此时出水溶解性氮的降低的主要原因在于微生物量/浓度的提高加快了底物利用速率[14]。在除磷方面,当除磷剂投加量不变时,对SRT≤15 d的系统除磷剂过量,因此出水磷酸盐浓度不变;但随着SRT升高导致系统中的总磷量增加,因此当SRT>15 d后药剂投加量相对不足,表现为出水磷酸盐浓度升高,同时由于磷抑制的降低,模拟结果中表现为PAOs菌组分的提升但不显著(仅0.1%)[20]。综合以上结果,当前系统最佳污泥龄宜控制在15~20 d,此时具有最佳脱氮除磷效果。
2.2.2 外回流比影响的量化分析
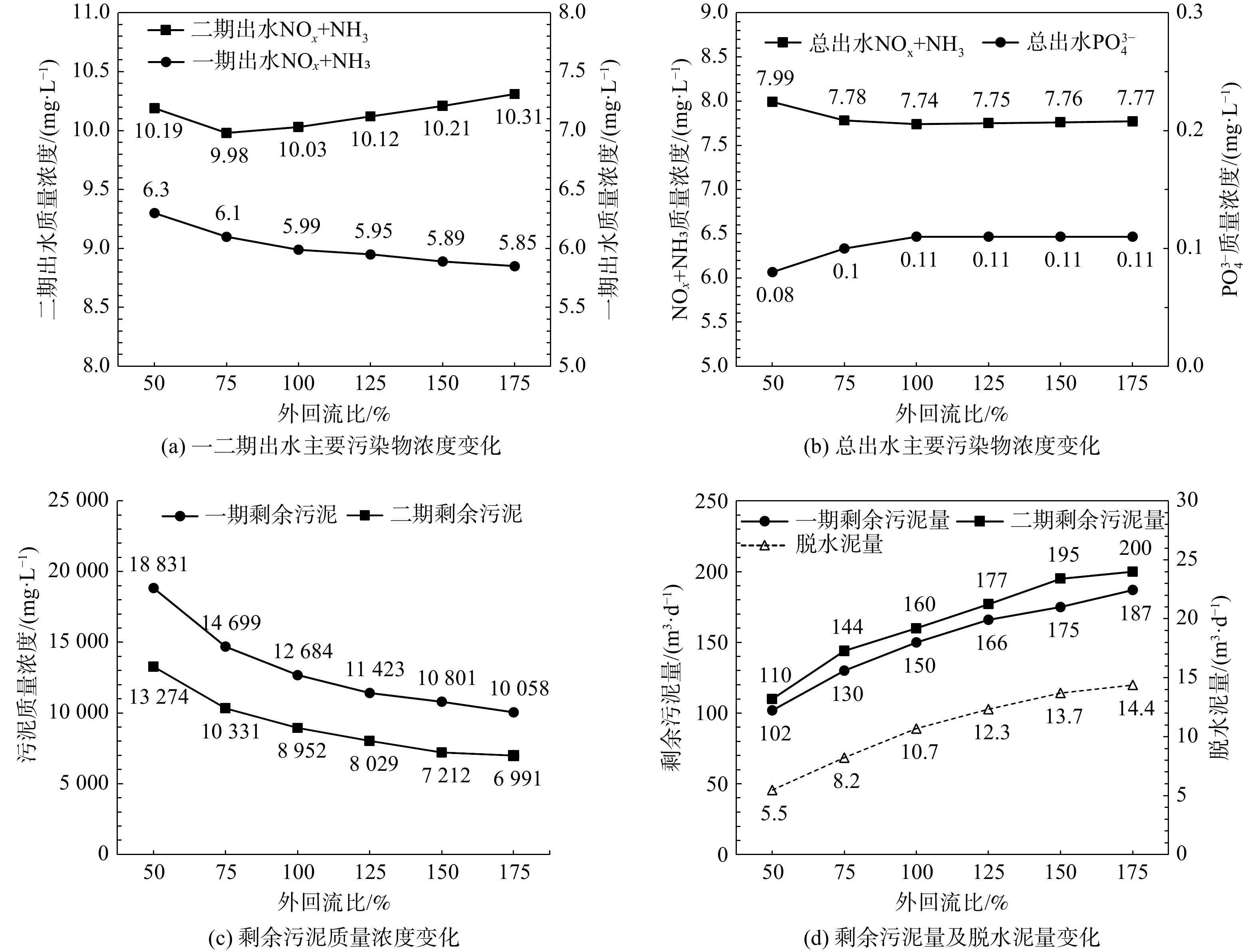
合理的外(污泥)回流比的控制可以有效发挥二沉池的澄清与污泥浓缩双重作用。因此,利用验证后的模型分别控制外回流比为50%、75%、100%、125%、150%、175%。在平均进水水质及正常运行工况下,稳态模拟不同外回流比时出水主要污染物浓度变化、剩余污泥浓度与泥量变化,结果见图3。图3(a)、图3(b)结果表明:随着外回流比增加,额外的硝酸氮与溶氧回流至系统前端,对活性污泥系统造成扰动但不显著;另一方面,随着外回流比的增加,二沉池的污泥浓缩作用被降低,导致二沉池底部污泥浓度降低,因此相同污泥龄时的排泥量增加,见图3(c)、图3(d)。综合以上结果,建议同时降低一二期污泥回流比至75%,此时对出水主要污染物浓度影响较小,并可有效增强二沉池浓缩功能。
2.2.3 内回流比影响的量化分析
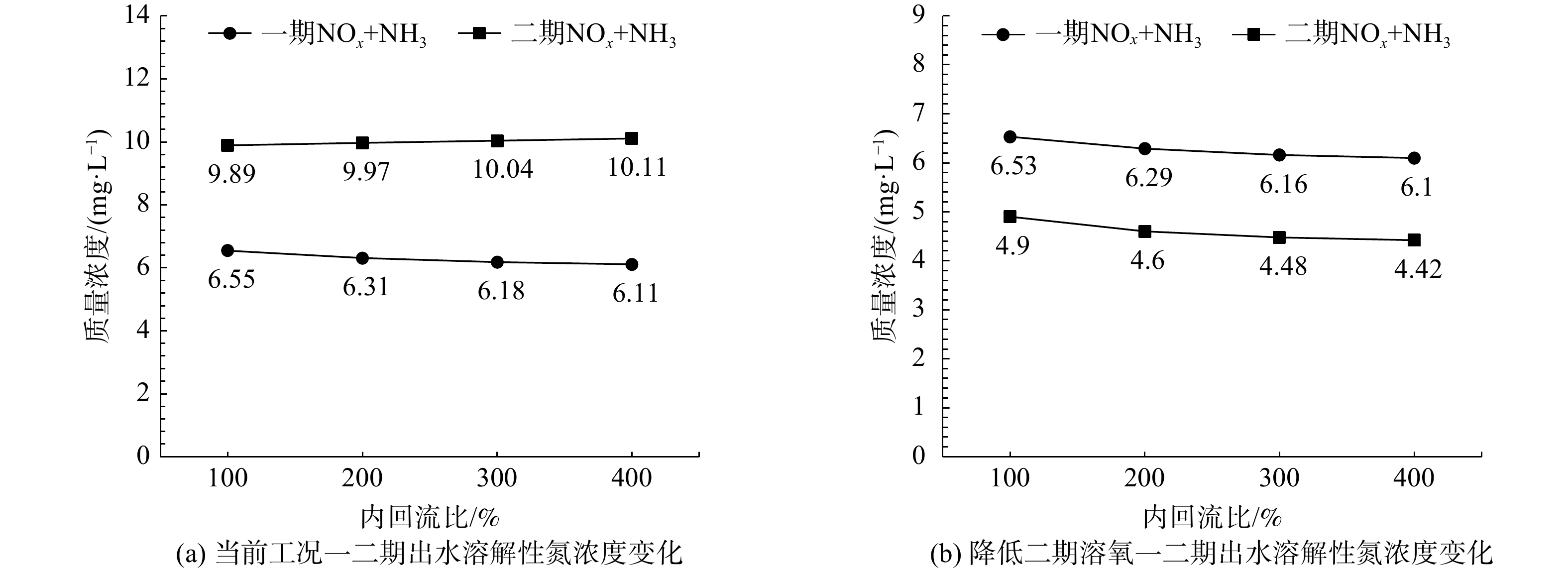
合理的内回流比控制可以最大化系统的反硝化脱氮能力。因此,利用验证后的模型分别模拟控制内回流比为100%、200%、300%、400%。基于平均进水水质,稳态模拟不同内回流比一、二期出水溶解性氮浓度,结果绘制于图4。图4(a)结果表明,当前工况增加内回流比对仅对一期脱氮效果有小幅提高,二期脱氮效果反而变差,这是由于该厂一、二期工艺内回流廊道紧邻1号表曝机(图1),并且二期表曝机功率高且水量分配少,造成沟内溶氧显著高于一期。因此,随着内回流比增加,回流溶氧对二期系统的扰动大于一期。如图4(b)结果所示,降低二期内回流点溶氧后,二期脱氮效果整体显著提高,同时增加内回流比后,一、二期均表现为脱氮效果的小幅提升。但整体而言,在回流溶氧的影响下,当前工况控制内回流无法显著提高系统脱氮能力。
2.2.4 沟内溶解氧影响的量化分析
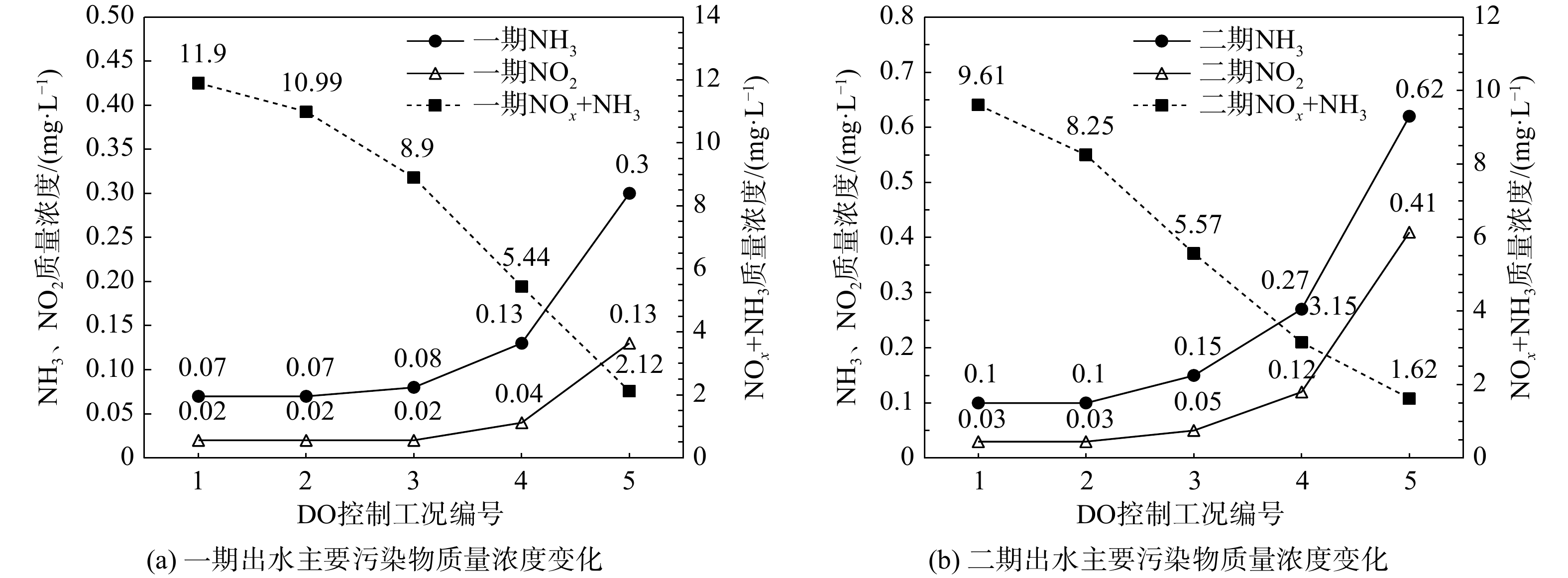
氧化沟工艺的溶氧(dissolved oxygen,DO)控制是制约系统脱氮能力的关键[14,17]。因此,对照图1,利用验证后的模型设计5种DO控制工况,控制氧化沟内3处表面曝气机DO分别为:工况1(1.5,2,2.5 mg·L−1)、工况2(1.5,2,2 mg·L−1)、工况3(1,1.5,2 mg·L−1)、工况4(1,1,1.5 mg·L−1)、工况5(0.5,1,1 mg·L−1)。基于平均进水水质,稳态模拟不同DO控制条件下一、二期出水氨氮(NH3)、亚硝氮(NO2−)及总溶解性氮浓度,结果绘制于图5。模拟结果表明:随着沟内DO浓度梯度的降低,一、二期工艺的脱氮效率均明显提升。这是由于降低DO浓度更有利于在氧化沟内形成缺氧区,强化同步硝化反硝化作用。但当DO≤0.5 mg·L−1时已接近NOB对DO的半饱和系数(表4),导致亚硝酸氮开始积累,因此,当前工况下DO梯度不宜低于工况5(0.5,1,1 mg·L−1)。
2.2.5 水温影响的量化分析
在实际污水处理过程中必须考虑水温对活性污泥系统的影响。基于控制变量法,上述结论均为模拟水温23 ℃时的模拟结果。为进一步确定优化参数,利用验证后的模型补充模拟了在低温(12 ℃)时不同SRT与不同氧化沟DO对出水水质的影响,结果绘制于图6。对比图2(a)及图5可发现:低温条件下污泥活性受到抑制,特别是对硝化细菌的影响较大,具体表现为在低SRT与低DO条件下,出水溶解性氮浓度显著升高。因此,在水温较低时(≤15℃),应适当提高SRT(20~25 d)并适当提高沟内DO,建议氧化沟三段溶氧不低于工况4(1,1,1.5 mg·L−1)。
2.3 方案实施与效果验证
基于技术分析与模拟优化结果,已识别出该厂工艺优化参数与调控方向。结合该厂设备运行情况及建模目标,提出的实施方案分如下2步。1)运行层面:调整一、二外回流泵由2台24 h运行减至1台24 h运行,即将回流比均降至约75%;调整一、二期排泥泵开启次数由4次减至3次,即将剩余污泥排放量减少约25%;2)控制层面:水温≤15 ℃时,控制两期污泥龄≥25 d(一期MLSS≈6 400 mg·L−1;二期MLSS≈5 300 mg·L−1),氧化沟内三处表曝机后端溶氧不低于工况5(0.5, 1, 1 mg·L−1);水温≥15℃时,控制两期污泥龄≥20 d(一期MLSS≈5 200 mg·L−1;二期MLSS≈4 400 mg·L−1),沟内溶氧不低于工况4(1, 1, 1.5 mg·L−1)。
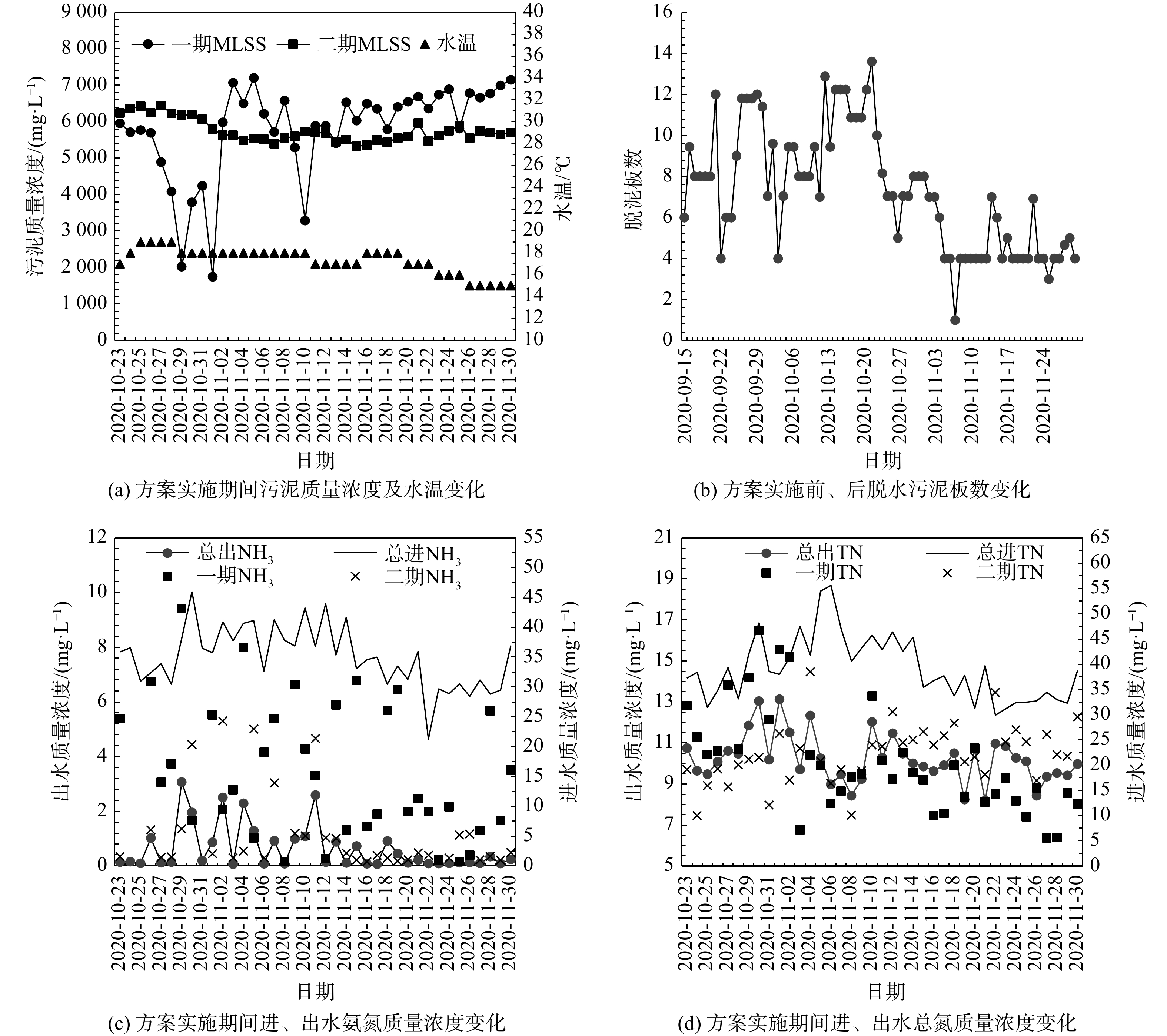
根据实施方案调整水厂的运行方式与操作参数,确保实施周期大于两期工艺的实际污泥龄(一期约22 d,二期约35 d)。最终连续跟踪水厂运行效果37 d,相关参数及水质指标变化见图7。由图7(a)可知,在方案实施初期,一期MLSS出现迅速下跌,二期MLSS则平稳下降,借助校正后的生物模型进行模拟分析后发现一期真实污泥回流比已降至不足40%,二期污泥回流比则稳定约75%。这是由于一期污泥回流泵的功率较低,当污泥回流比降低导致剩余污泥浓度增加后,一台泵单独工作不能实现稳定回流控制[21]。稳定后,一期改为1台泵24 h连续运行、1台泵运行8 h·d−1,二期则稳定降至1台泵24 h运行,但该方式导致一期MLSS的波动幅度明显,因此,建议该厂远期可更换功率与二期一致的污泥回流泵。
配合污泥回流比的调整,两期剩余污泥排放量减少约25%后,亦间接增加了二沉池污泥浓缩时间。由图7(b)可知,脱泥板数由优化前(2020-09-15—2020-10-22)的平均每日9.4板,减少至优化后(2020-10-23—2020-11-30)平均每日5.1板。统计结果表明,优化后的污泥脱水药剂(CaO、FeCl3)用量较优化前节省近46%;污泥回流泵、剩余污泥泵、板框压滤机等合计电耗节约840 kW·h·d−1。因调试期间水温已持续降低至15 ℃,因此,通过污泥回流比及排泥量的调整,可成功将一期污泥龄由22 d增至28 d,二期污泥龄由35 d降至29 d。
在溶解氧控制方面,因一期氧化沟三处表曝机后端溶氧(0.8,1,1.1 mg·L−1)已十分接近优化方案控制工况5(0.5,1,1 mg·L−1),因此,方案实施期间主要降低二期过高溶氧(1.7,2,2.4 mg·L−1),操作方式为分别减少3台表曝机启/闭时间。由图7(c)、图7 (d)可知,在进水水质波动不大的情况下,方案实施初期(2020-10-23—2020-11-11)通过降低二期溶氧至(0.8,1.1,1.4 mg·L−1),导致二期出水氨氮增加(NH3,二期EFF≈1.9 mg·L−1),但二期出水总氮(TN,二期EFF≈9.8 mg·L−1)显著低于一期(TN,一期EFF≈11.5 mg·L−1)。然而,受设备及仪表问题限制,需通过手动调整3台表曝机启/闭时间且需手动定时测量DO才可实现对溶氧的控制,这极大增加了运行班组工作强度。因此,在实施后期(2020-11-12—2020-11-30)取消了对溶氧的人工干预,二期平均溶氧恢复至控制前水平(1.5,2.2,2.5 mg·L−1),此时二期出水氨氮恢复低值(NH3,二期EFF≈0.5 mg·L−1)且出水总氮(TN,二期EFF≈11.1 mg·L−1)开始高于一期出水(TN,一期EFF≈8.5 mg·L−1),该现象与模拟结论完全吻合。因此,建议该厂远期增加在线仪表维护,提高自动化水平。
3. 结论
1)得益于可靠的生物建模技术,污水处理厂的边界条件和真实运行参数得到评估与修正。利用校正后的生物模型不仅可以实现对整条工艺线中各点水质、水量的全部量化,还可以模拟对比不同工况对活性污泥系统的影响,完成对水厂运行问题的梳理与诊断,并最终确定理想状态的最佳优化参数。
2)案例污水厂的优化实施效果表明,校正后的模型正确识别了该厂常规工况下的运行问题,基于生物模型提出的运行优化方案切实可行。将模型与应用相结合,不仅解决了案例厂脱水剂药耗过高问题,实现节能降耗,还确定了污水处理厂的远期控制策略与优化方向,亦证明了生物建模在污水处理厂运营问题诊断与运行优化的指导价值。
-

 点击查看大图
点击查看大图
计量
- 文章访问数: 2443
- HTML全文浏览数: 1211
- PDF下载数: 1125
- 施引文献: 0





 下载:
下载: